Welcome to Iris Biotech
For better service please confirm your country and language we detected.

For better service please confirm your country and language we detected.

Thank you very much for your interest in our products. All prices listed on our website are ex-works, Germany, and may attract customs duties when imported.
You may/will be contacted by the shipping company for additional documentation that may be required by the US Customs for clearance.
We offer you the convenience of buying through a local partner, Peptide Solutions LLC who can import the shipment as well as prepay the customs duties and brokerage on your behalf and provide the convenience of a domestic sale.
Continue to Iris Biotech GmbHSend request to US distributorPublished on 02.12.2016
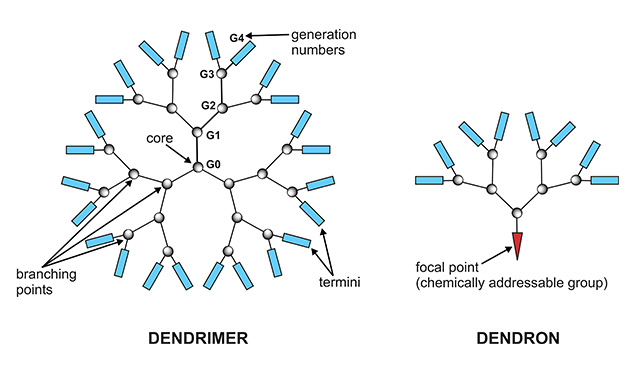
Dendronization is a synthetic method by which functional materials with specific architectures and useful new properties can be created.
We now offer several lysine dendrons equipped with a hydrazide functional group at the focal point/C-terminus for chemoselective conjugation to molecules bearing carbonyl groups. The conjugation can be performed with (or without) subsequent reduction of the hydrazone bond by NaBH4.
For your convenience, we offer a selection of Lys dendrons with spacer arms of different lengths (LS-3610, LS-3620, LS-3630). The terminal lysine residues can be further functionalized with amino-reactive molecules.
Moreover, we also offer a lysine dendron bearing several serine residues (LS-3640). By selective conjugation of this building block to peptides or polymers using the hydrazone ligation technique, multiple Ser residues can be incorporated into the target molecule, thus multiplying the number of reactive functional groups. Furthermore, these Ser residues can be converted to glyoxylic acid by periodate oxidation with NaIO4, thus creating multiple attachment sites for payloads with aldehyde-reactive groups.
A further variant of our new dendrons is a DOTA-functionalized derivative (LS-3600). Conjugation of this dendron to peptide or polymer carrier molecules facilitates delivery of paramagnetic ions (Gd3+; Mn2+) or radioactive isotopes. Applications include the targeted delivery of MR contrast agents (e.g. Gd3+) and radioimmunotherapy.
Lastly, we included a triphenylphosphonium (TPP)-bearing dendron into our portfolio (LS-3650). This type of building block was designed for the targeted delivery of peptides, polymers and different drugs to mitochondria.
Our new dendrons (in the form of hydrazides) can be applied for the modification of synthetic or natural polymers containing reactive aldehyde groups (peptides must first be modified by oxidation of Ser or Thr residues). The dendrons can for example be applied for the design of gene delivery systems or for modifying the surface properties of polymers. The conjugation process can be performed using the hydrazone ligation approach with (or without) subsequent reduction of the resulting hydrazone bond by NaBH4. We can provide all necessary protocols.
A dendronized heparin-gadolinium polymer self-assembled into a nanoscale system as a potential magnetic resonance imaging contrast agent; C. Guo, L. Sun, W. She, N. Li, L. Jiang, K. Luo, Q. Gong and Z. Gu; Polymer Chemistry 2016; 7: 2531-2541. doi:10.1039/c6py00059b
Effect of the Conjugation Density of Triphenylphosphonium Cation on the Mitochondrial Targeting of Poly(amidoamine) Dendrimers; E. R. Bielski, Q. Zhong, M. Brown and S. R. P. da Rocha; Molecular pharmaceutics 2015; 12: 3043-3053. doi:10.1021/acs.molpharmaceut.5b00320
Mitochondrial targeting dendrimer allows efficient and safe gene delivery; X. Wang, N. Shao, Q. Zhang and Y. Cheng; Journal of Materials Chemistry B 2014; 2: 2546-2553. doi:10.1039/c3tb21348j
A hydrazide-anchored dendron scaffold for chemoselective ligation strategies; L. O'Donovan and P. A. De Bank; Organic & Biomolecular Chemistry 2014; 12: 7290-7296. doi:10.1039/c4ob00870g
Peptide targeted tripod macrocyclic Gd(III) chelates for cancer molecular MRI; Z. Zhou, X. Wu, A. Kresak, M. Griswold and Z.-R. Lu; Biomaterials 2013; 34: 7683-7693. doi:http://dx.doi.org/10.1016/j.biomaterials.2013.06.057
Synthesis and Evaluation of a Peptide Targeted Small Molecular Gd-DOTA Monoamide Conjugate for MR Molecular Imaging of Prostate Cancer; X. Wu, S. M. Burden-Gulley, G.-P. Yu, M. Tan, D. Lindner, S. M. Brady-Kalnay and Z.-R. Lu; Bioconjugate Chemistry 2012; 23: 1548-1556. doi:10.1021/bc300009t
Dendronization: A Useful Synthetic Strategy to Prepare Multifunctional Materials; J. I. Paez, M. Martinelli, V. Brunetti and M. C. Strumia; Polymers 2012; 4: 355.
Surface conjugation of triphenylphosphonium to target poly(amidoamine) dendrimers to mitochondria; S. Biswas, N. S. Dodwadkar, A. Piroyan and V. P. Torchilin; Biomaterials 2012; 33: 4773-4782. doi:http://dx.doi.org/10.1016/j.biomaterials.2012.03.032
Multiple Triphenylphosphonium Cations as a Platform for the Delivery of a Pro-Apoptotic Peptide; N. Kolevzon, U. Kuflik, M. Shmuel, S. Benhamron, I. Ringel and E. Yavin; Pharmaceutical Research 2011; 28: 2780. doi:10.1007/s11095-011-0494-6
MRI Detection of VEGFR2 in Vivo Using a Low Molecular Weight Peptoid−(Gd)8-Dendron for Targeting; L. M. De León-Rodríguez, A. Lubag, D. G. Udugamasooriya, B. Proneth, R. A. Brekken, X. Sun, T. Kodadek and A. Dean Sherry; Journal of the American Chemical Society 2010; 132: 12829-12831. doi:10.1021/ja105563a
Multiple Triphenylphosphonium Cations Shuttle a Hydrophilic Peptide into Mitochondria; S. E. Abu-Gosh, N. Kolvazon, B. Tirosh, I. Ringel and E. Yavin; Molecular pharmaceutics 2009; 6: 1138-1144. doi:10.1021/mp900032r
Inhibition of Mitosis by Glycopeptide Dendrimer Conjugates of Colchicine; D. Lagnoux, T. Darbre, M. L. Schmitz and J.-L. Reymond; Chemistry – A European Journal 2005; 11: 3941-3950. doi:10.1002/chem.200401294
Synthesis and Application of Peptide Dendrimers As Protein Mimetics; J. P. Tam and J. C. Spetzler; Current Protocols in Immunology 2001. doi:10.1002/0471142735.im0906s34
Assembly of cyclic peptide dendrimers from unprotected linear building blocks in aqueous solution; T. D. Pallin and J. P. Tam; Chemical Communications 1996: 1345-1346. doi:10.1039/cc9960001345