Welcome to Iris Biotech
For better service please confirm your country and language we detected.

For better service please confirm your country and language we detected.

Thank you very much for your interest in our products. All prices listed on our website are ex-works, Germany, and may attract customs duties when imported.
You may/will be contacted by the shipping company for additional documentation that may be required by the US Customs for clearance.
We offer you the convenience of buying through a local partner, Peptide Solutions LLC who can import the shipment as well as prepay the customs duties and brokerage on your behalf and provide the convenience of a domestic sale.
Continue to Iris Biotech GmbHSend request to US distributorPublished on 30/11/2022

Typically, peptides are synthesized via solid phase peptide synthesis (SPPS). Its main advantage compared to classical solution phase synthesis is the fast and easy separation of the desired product from excess reagents by filtration. Nevertheless, especially for long sequences, the probability that difficult fragments are appearing is rather high, which will lower coupling efficiencies, lead to deletion sequences, and lower overall yields. Consequently, different ligation techniques have been developed to facilitate the access to longer peptide sequences and proteins.
Native chemical ligation (NCL) of unprotected peptide segments involves the reaction between a first peptide fragment alpha-thioester and a second peptide fragment, which carries a cysteine on the N-terminus, to yield a product with a native amide bond at the ligation site after desulfurization. As Cysteines (Cys) appear very rarely in natural sequences, are prone to racemization, and show slow kinetics in the ligation reaction, chemists expanded the native chemical ligation toolbox and developed alternative protocols.
➔ Interested in the formation of peptide thioesters? Read our blog post about Dawson linkers!
The key advantage of carrying out ligation chemistry with selenoamino acids rather than thioamino acids is that chemoselective deselenization can be performed under mild conditions that do not affect unprotected Cys residues. After oxidative deselenization, a Serine in place of Selenocysteine results at the ligation junction.

Sulfur- vs. selenium-based chemical ligation.
Our portfolio contains a variety of selenocysteine (Sec) derivatives as well as selenazolidine carboxylic acids (Sez derivatives). Sez can be deprotected and converted to Sec by treatment with O‑methylhydroxylamine (MeONH2) at pH 4 or by using Cu(II) salts.
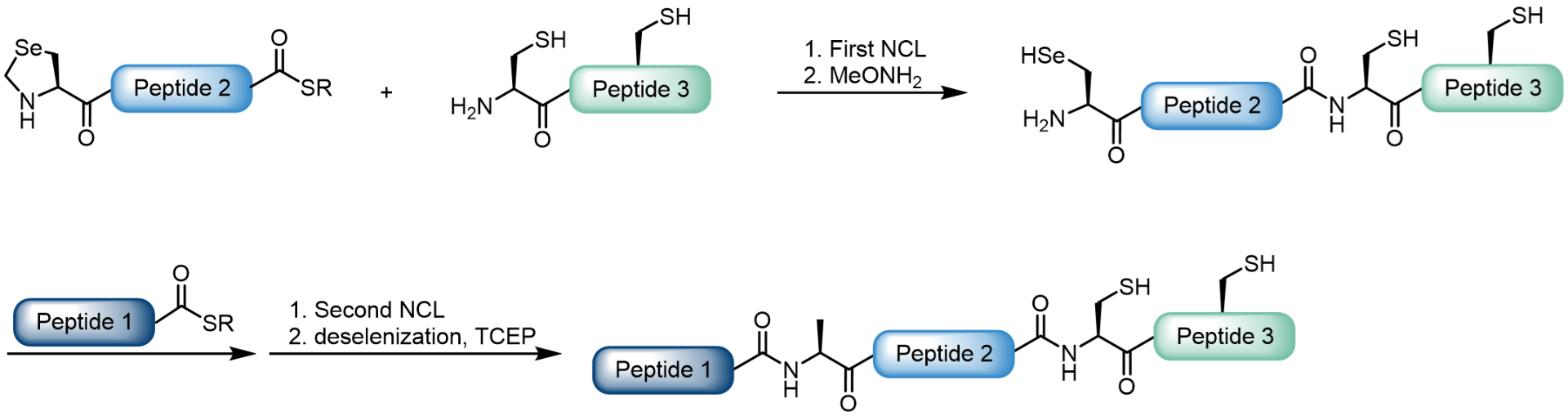
Usage of a Sez building block for native chemical ligation.
➔ Check out the related products at the bottom of this side to see available Sec and Sez derivatives!
Besides NCL, other ligation technologies have gained increasing interest, e.g. hydrazone ligation. Peptide hydrazides can be easily synthesized using hydrazone resins, which are polystyrene resins functionalized with the hydrazone linker. The hydrazone linker is completely stable in the course of standard Fmoc-SPPS. Final application of cleavage cocktails containing neat TFA permits to obtain the desired peptide hydrazide in high yields and purity.
The synthesized peptide hydrazides can be conjugated to an aldehyde or ketone of a second peptide fragment. The resulting conjugate shows a non-natural hydrazone linkage replacing the natural amide bond. This, however, is in many cases tolerated, and the conjugate displays the same properties as a natural protein fragment.

Schematic illustration of the hydrazone ligation.
Besides, peptide hydrazides can also be converted to peptide thioesters and be used for subsequent NCL. Two different protocols have been published recently.

Method 1: Synthesis of peptide thioesters from peptide hydrazides via azide formation.

Method 2: Synthesis of peptide thioesters from peptide hydrazides using acetylacetone.
Another strategy for the generation of large peptides and proteins is the so-called activated cysteine-based protein ligation (ACPL). This method is based on the direct activation of Cysteine by the small molecule cyanylating reagent 2-nitro-5-thiocyanatobenzoic acid (NTCB). The transferred cyanide then forms a thiocyanate which undergoes a reversible intramolecular addition with the Cys N-amide to generate a five-membered 1-acyl-2-iminothiazolidine intermediate which can be reacted with nucleophiles. When the nucleophile is hydrazine, the afforded peptide hydrazide can then undergo ligation, either via reaction with an aldehyde/ketone (hydrazone ligation) or via transformation to a peptide thioester and subsequent chemical ligation, both as described above. Without addition of a nucleophilic amine, the formed five-membered ring undergoes hydrolysis.
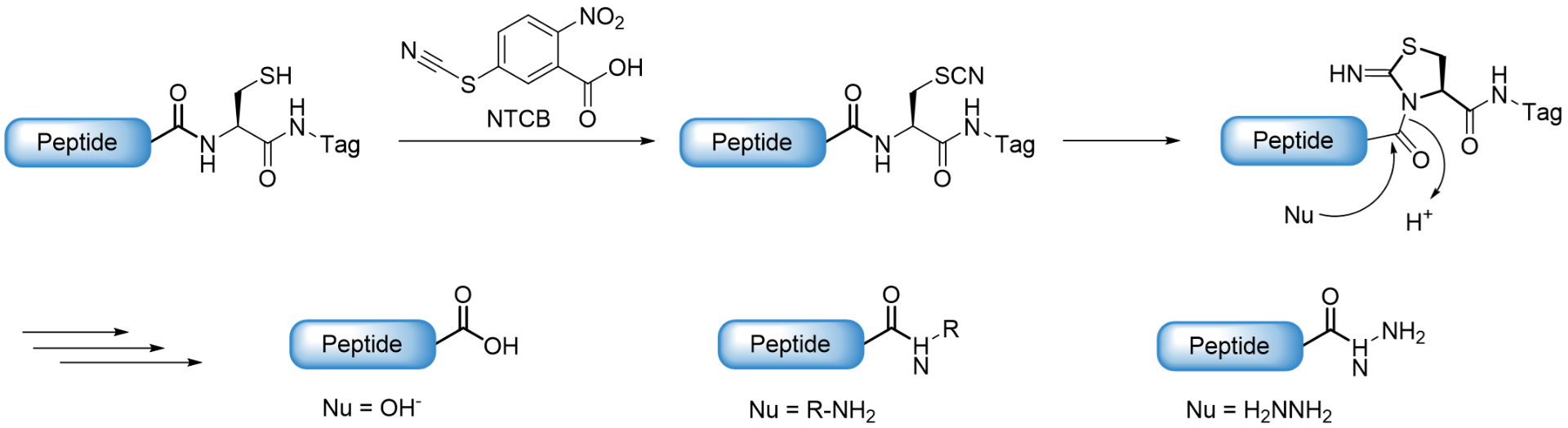
Schematic illustration of the activated Cysteine-based protein ligation.
In addition, the formed S-cyano-Cysteine with ist highly labile S-Cbeta bond can be triggered for the generation of Dehydroalanine (Dha) via an E2 elimination reaction. Especially if Cysteine is at the flexible C-terminal end of a protein, the formation of Dehydroalanine is highly effective. Dha contains an alpha,beta-unsaturated carbonyl moiety that can undergo both Michael addition and cross coupling reactions to generate various post-translational modifications or their analogues.
The typically used methods for the conversion of Cys to Dha in a protein require rather harsh conditions not compatible with native proteins. NTCB allows the transformation of Cys in a protein to Dha under bio-orthogonal conditions at pH 7.
➔ You need more details about ligation technologies? Download our brochure!
References:
Expressed Protein Ligation without Intein; Y. Qiao, G. Yu, K. C. Kratch, X. Aria Wang, W. Wei Wang, S. Z. Leeuwon, S. Xu, J. S. Morse, W. Ray Liu; J. Am. Chem. Soc. 2002; 142: 7057-7054. https://doi.org/10.1021/jacs.0c00252
Site-Specific Conversion of Cysteine in a Protein to Dehydroalanine Using 2-Nitro-5-thiocyanatobenzoic Acid; Y. Qiao, G. Yu, S. Z. Leeuwon, W. Ray Liu; Molecules 2021; 26: 2619. https://doi.org/10.3390/molecules26092619
Accelerated Protein Synthesis via One-Pot Ligation-Deselenization Chemistry; N. J. Mitchell, J. Sayers, S. S. Kulkarni, D. Clayton, A. M. Goldys, J. Ripoll-Rozada, P. J. Barbosa Pereira, B. Chan, L. Radom, R. J. Payne; Chem 2017; 2: 703-715. https://doi.org/10.1016/j.chempr.2017.04.003
Oxidative Deselenization of Selenocysteine: Applications for programmed Ligation at Serine; L. R. Malins, N. J. Mitchell, S. McGowan, R. J. Payne; Angew. Chem. Int. Ed. Engl. 2015; 54: 12716-12721. https://doi.org/10.1002/anie.201504639
Chemical Synthesis of Proteins with Non-Strategically Placed Cysteines Using Selenazolidine and Selective Deselenization; P. S. Reddy, S. Dery, N. Metanis; Angew. Chem. Int. Ed. 2016; 55(3): 992-995. https://doi.org/10.1002/anie.201509378
Copper-Mediated Deprotection of Thiazolidine and Selenazolidine Derivatives Applied to Native Chemical Ligation; N. Naruse, D. Kobayashi, K. Ohkawachi, A. Shigenaga, A. Otaka; J. Org. Chem. 2020; 85(3): 1425-1433. https://doi.org/10.1021/acs.joc.9b02388
Chemical synthesis of proteins using peptide hydrazides as thioester surrogates; J. S. Zheng, S. Tang, Y. K. Qi, Z. P. Wang, L. Liu; Nat Protoc 2013; 8: 2483-2495. https://doi.org/10.1038/nprot.2013.152
Convenient method of peptide hydrazide synthesis using a new hydrazone resin; P. S. Chelushkin, K. V. Polyanichko, M. V. Leko, M. Y. Dorosh, T. Bruckdorfer, S. V. Burov; Tetrahedron Letters 2015; 56: 619-622. https://doi.org/10.1016/j.tetlet.2014.12.056
Leveraging the Knorr Pyrazole Synthesis for the Facile Generation of Thioester Surrogates for use in Native Chemical Ligation; D. T. Flood, J. C. J. Hintzen, M. J. Bird, P. A. Cistrone, J. S. Chen, P. E. Dawson; Angew Chem Int Ed Engl 2018; 57: 11634-11639. https://doi.org/10.1002/anie.201805191
Protein chemical synthesis by ligation of peptide hydrazides; G. M. Fang, Y. M. Li, F. Shen, Y. C. Huang, J. B. Li, Y. Lin, H. K. Cui, L. Liu; Angew Chem Int Ed Engl 2011; 50: 7645-7649. https://doi.org/10.1002/anie.201100996