Welcome to Iris Biotech
For better service please confirm your country and language we detected.

For better service please confirm your country and language we detected.

Thank you very much for your interest in our products. All prices listed on our website are ex-works, Germany, and may attract customs duties when imported.
You may/will be contacted by the shipping company for additional documentation that may be required by the US Customs for clearance.
We offer you the convenience of buying through a local partner, Peptide Solutions LLC who can import the shipment as well as prepay the customs duties and brokerage on your behalf and provide the convenience of a domestic sale.
Continue to Iris Biotech GmbHSend request to US distributorPublished on 20/04/2021
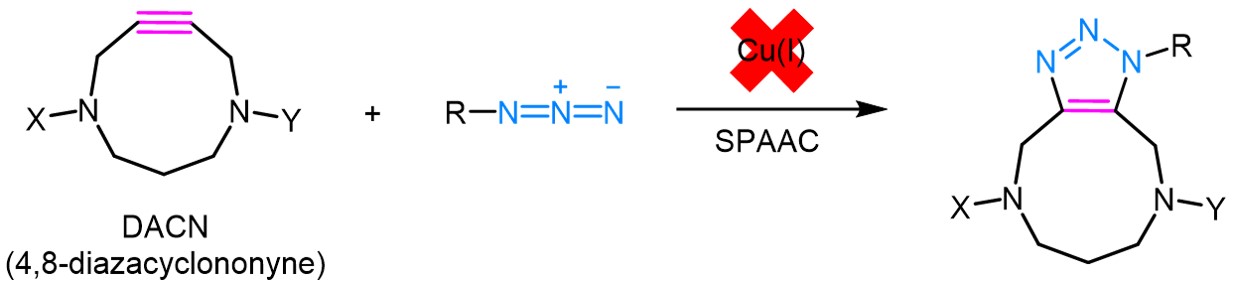
The alkyne moiety represents one of the most versatile units in organic chemistry. Its reactivity can be finetuned and well-adjusted depending on surrounding heteroatoms and/or the steric and electronic demand of its substituents. One of the most prominent reactions of alkynes is the so-called Click reaction, which has developed to a widely used conjugation technique. In this context, as realized by Wittig and Krebs in 1961, cyclic alkynes benefit of remarkable high reactivities compared to their linear, unstrained counterparts, which show slow kinetics in Huisgen 1,3-dipolar cycloadditions with azides.
Despite the potential acceleration of the latter mentioned reaction by copper catalysis the use of this metal drastically limits in vivo applications e.g. due to high cell toxicity and undesired oxidation of proteins.
Thus, the so-called strain-promoted azide-alkyne cycloaddition (SPAAC) is widely utilized as copper-free click reaction in a broad range of research fields. However, when using cyclooctynes, their high reactivity comes with the drawback of causing thermal decomposition and/or oligomerization during storage and experimental handling. Furthermore, competitively occurring addition reaction with biological thiols, e.g. cysteine residues in proteins or peptides, diminish their biorthogonality.
In contrast, the relatively new developed 4,8-diazacyclononynes (DACNs) possess high thermal and chemical stability while exhibiting a reactivity twice as high as OCT and benefiting of increased hydrophilicity and high selectivity towards ynophiles. Furthermore, the embedded endocyclic nitrogen atoms can be utilized as connectors for further conjugation thus making the compound functionally versatile.
Within our portfolio, we offer a variety of substituted 4,8-diazacyclononynes which are listed at the end of this article under “related products”. Furthermore, our portfolio includes a variety of azide-derivatized amino acids and building blocks and many more are available via our Custom Synthesis Service.
References:
A strain-promoted [3 + 2] azide-alkyne cycloaddition for covalent modification of biomolecules in living systems; N. J. Agard, J. A. Prescher and C. R. Bertozzi; J Am Chem Soc 2004; 126: 15046-7. https://doi.org/10.1021/ja044996f
Bioconjugation with strained alkenes and alkynes; M. F. Debets, S. S. van Berkel, J. Dommerholt, A. T. Dirks, F. P. Rutjes and F. L. van Delft; Acc Chem Res 2011; 44: 805-15. https://doi.org/10.1021/ar200059z
Strain-Promoted 1,3-Dipolar Cycloaddition of Cycloalkynes and Organic Azides; J. Dommerholt, F. Rutjes and F. L. van Delft; Top Curr Chem (Cham) 2016; 374: 16. https://doi.org/10.1007/s41061-016-0016-4
Selective strain-promoted azide-alkyne cycloadditions through transient protection of bicyclo[6.1.0]nonynes with silver or gold; K. Adachi, T. Meguro, Y. Sakata, K. Igawa, K. Tomooka, T. Hosoya and S. Yoshida; Chem Commun (Camb) 2020; 56: 9823-9826. https://doi.org/10.1039/d0cc04606j
Copper-Free Huisgen Cycloaddition for the 14-3-3-Templated Synthesis of Fusicoccin-Peptide Conjugates; R. Masuda, Y. Kawasaki, K. Igawa, Y. Manabe, H. Fujii, N. Kato, K. Tomooka and J. Ohkanda; Chem Asian J 2020; 15: 742-747. https://doi.org/10.1002/asia.202000042
Heteroatom-embedded Medium-Sized Cycloalkynes: Concise Synthesis, Structural Analysis, and Reactions; R. Ni, N. Mitsuda, T. Kashiwagi, K. Igawa, K. Tomooka; Angew. Chem. Int. Ed. 2015; 54: 1190-1194. https://doi.org/10.1002/anie.201409910.
Thieme Chemistry Journals Awardees: Where Are They Now? One-Pot Synthesis of Versatile Buckle Units for Click Chemistry: 4,8-Diazacyclononynes (DACNs); K. Igawa, S. Aoyama, Y. Kawasaki, T. Kashiwagi, Y. Seto, R. Ni, N. Mitsuda, K. Tomooka; Synlett 2017; 28: 2110-2114. https://doi.org/10.1055/s-0036-1588839.