Welcome to Iris Biotech
For better service please confirm your country and language we detected.

For better service please confirm your country and language we detected.

Thank you very much for your interest in our products. All prices listed on our website are ex-works, Germany, and may attract customs duties when imported.
You may/will be contacted by the shipping company for additional documentation that may be required by the US Customs for clearance.
We offer you the convenience of buying through a local partner, Peptide Solutions LLC who can import the shipment as well as prepay the customs duties and brokerage on your behalf and provide the convenience of a domestic sale.
Continue to Iris Biotech GmbHSend request to US distributorPublished on 25/01/2022
For the development of innovative therapies and new drug agents, detailed information to understand a drug’s metabolic and pharmacokinetic pathway, its distribution and potential accumulation and toxicities are of crucial importance. The so-called ADMET profile, the absorption, distribution, metabolism, excretion, and toxicology of a drug, is one major checkpoint during drug discovery.
During such analysis not only the drug and drug-derived agents are monitored, but also various endogenous metabolites, which change rapidly in response to physiological perturbations such as drug consumption, as they serve as reporters to monitor the response to drug treatment.
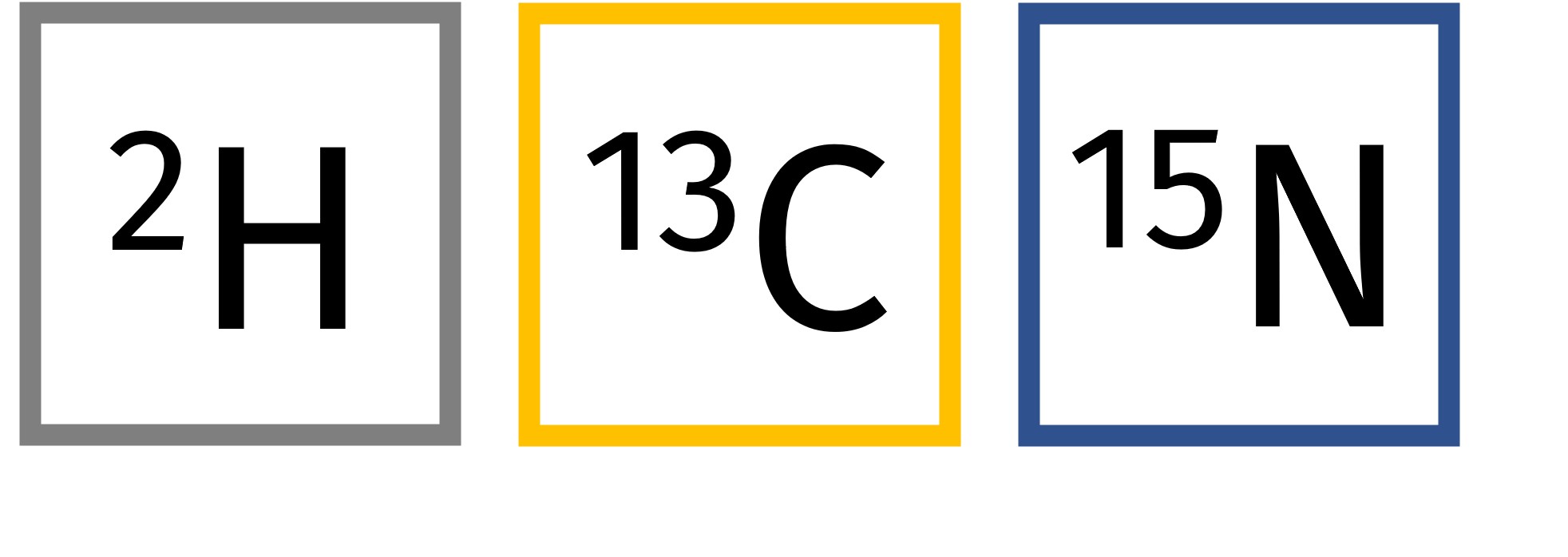
In this regard, quantitative metabolomics is a frequently utilized tool for the identification of such biomarkers among other metabolites. In the past, radioisotopes have been utilized for tracking due to their sensitive detectability. However, nowadays, the use of stable isotopes has increased due to greater availability and safety. Some of the most common stable isotopes are 2H, 13C, and 15N with natural abundances of 0.015%, 1.10%, and 0.366%. Thereby, quantification of metabolite levels via mass spectrometry is frequently employed.
Typically, the heavy labeled standards are used as internal standards for quantitative metabolite profiling, allowing accurate measurements and diagnosis. In differential proteomics, two samples are compared, one labeled with the naturally occurring isotope abundance (light) and the other with a stable isotope of low natural abundance (heavy). The samples are mixed, and their ratio is analyzed by mass spectrometry to determine the relative amounts of a given substance in two samples. Such results are unaffected by errors during sample preparation and instrument response.
Iris Biotech is presenting a selection of stable isotopically labeled (SIL) derivatives to be used as reference standard for analysis.
➔ Interested? Scroll down to the section “related products” to see all available drug derivatives.
References:
Isotopic labeling of metabolites in drug discovery applications; J. Iglesias, L. Sleno, D. A. Volmer; Curr. Drug Metab. 2012; 13(9): 1213-1225. https://doi.org/10.2174/138920012803341357.
The use of stable isotope labelling for the analytical chemistry of drugs; C. J. Unkefer, R. A. Martinez; Drug Test Anal. 2012; 4(3-4): 303-7. https://doi.org/10.1002/dta.361.
Chapter 25 The Use of Isotopically Labeled Compounds in Drug Discovery; C. S. Elmore; Annual Reports in Medicinal Chemistry 2009; 44: 515-534. https://doi.org/10.1016/S0065-7743(09)04425-X.
Stable isotope labeling – Liquid chromatography/mass spectrometry for quantitative analysis of androgenic and progestagenic steroids; N. Guo, P. Liu, J. Ding, S.-J. Zheng, B.-F. Yuan, Y.-Q. Feng; Anal. Chim. Acta 2016; 905: 106-114. https://doi.org/10.1016/j.aca.2015.12.010.
Stable isotope labelling methods in mass spectrometry-based quantitative proteomics; O. Chahrour, D. Cobice, J. Malone; J. Pharm. Biomed Anal. 2015; 113(2): 2-20. https://doi.org/10.1016/j.jpba.2015.04.013.
Stable isotopic labeling of proteins for quantitative proteomic applications; G. W. Becker; Briefings in Functional Genomics 2008; 7(5): 371-382. https://doi.org/10.1093/bfgp/eln047.