Welcome to Iris Biotech
For better service please confirm your country and language we detected.

For better service please confirm your country and language we detected.

Thank you very much for your interest in our products. All prices listed on our website are ex-works, Germany, and may attract customs duties when imported.
You may/will be contacted by the shipping company for additional documentation that may be required by the US Customs for clearance.
We offer you the convenience of buying through a local partner, Peptide Solutions LLC who can import the shipment as well as prepay the customs duties and brokerage on your behalf and provide the convenience of a domestic sale.
Continue to Iris Biotech GmbHSend request to US distributorPublished on 22/02/2023

Post-translational protein modification (PTM) is widespread in nature and desirable in most cases. Typically, these modifications are targeted and enzyme-mediated, for example phosphorylation, glycosylation, or farnesylation.
However, there are also non-enzymatic modifications that proteins undergo (nPTM), e.g. through aging, various diseases such as diabetes or cancer, or exposure to heat. These are often unwanted reactions with metabolic compounds. For example, surface exposed Lysine or Arginine side chains can react with the metabolite methylglyoxal to finally form Carboxyethyllysine (CEL), Methylglyoxal-hydroimidazolone (MG-H1), or Argpyrimidine (Apy):
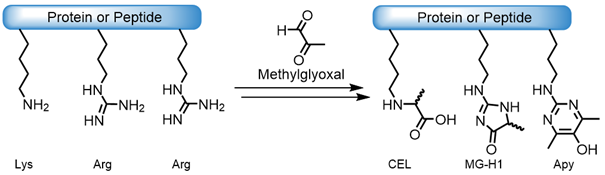
Reaction of surface exposed Lys-/Arg-side chains with methylglyoxal.
Apy has – for example – been detected in aged and cataract-affected eye lenses, in amyloidotic polyneuropathy patients’ amyloid fibers, or in cells and tissues from diabetic and cancer patients.
To elucidate the biological effects of such Apy formation, it is important to introduce Argpyrimidine quantitatively at a defined position in a peptide or protein of interest. This can best be achieved by solid phase peptide synthesis (SPPS). To be applicable in typical SPPS conditions, such an Apy building block should have an Fmoc-protection on the α-amine and side chain protections with acid-labile protecting groups.
The combination of sidechain protecting groups not only has to satisfy the condition of orthogonality to typical Fmoc synthesis conditions, but also must be synthetically accessible in somehow reasonable yields. After several attempts, Professor Christian F. W. Becker and his team at the University of Vienna developed the building block Fmoc-Argpyrimidine(Pbf,TBMS)-OH (FAA5530), which can easily be used in standard SPPS protocols and is available from Iris Biotech:

Chemical structure of Fmoc-Argpyrimidine(Pbf,TBMS)-OH available at Iris Biotech.
To introduce Argpyrimidine into bigger proteins, a semi-synthetic approach can be applied:
(I) Production of the largest part(s) of the protein by recombinant synthesis,
(II) SPPS of the the Apy-containing fragment,
(III) Fragment condensation to the final full-length protein.
If you want to learn more about the interesting journey how to produce proteins with non-enzymatic post-translational modifications (nPTM), you should follow our workshop with Prof. Becker:
https://www.iris-biotech.de/workshop
Several other modified amino acids of this kind are listed in our Flyer “Maillard and Amadori Reaction Products”, either as free amino acids, or labeled with Deuterium for mass spectrometry or as multifold protected building blocks for SPPS.
References:
Synthetic Approach to Argpyrimidine as a Tool for Investigating Nonenzymatic Posttranslational Modification of Proteins; M. Matveenko, C. F. W. Becker; Synlett 2017; 28: 1950-1955. https://doi.org/10.1055/s-0036-1588225
Impaired Chaperone Activity of Human Heat Shock Protein Hsp27 Site-Specifically Modified with Argpyrimidine; M. Matveenko, E. Cichero, P. Fossa, C. F. W. Becker; Ang. Chem. Int. Ed. 2016; 55: 11397-11402. https://doi.org/10.1002/anie.201605366
Random coil shifts of posttranslationally modified amino acids; A. C. Conibear, K. J. Rosengren, C. F. W. Becker, H. Kaehlig; Journal of Biomolecular NMR 2019; 73: 587-599. https://doi.org/10.1007/s10858-019-00270-4