Welcome to Iris Biotech
For better service please confirm your country and language we detected.

For better service please confirm your country and language we detected.

Thank you very much for your interest in our products. All prices listed on our website are ex-works, Germany, and may attract customs duties when imported.
You may/will be contacted by the shipping company for additional documentation that may be required by the US Customs for clearance.
We offer you the convenience of buying through a local partner, Peptide Solutions LLC who can import the shipment as well as prepay the customs duties and brokerage on your behalf and provide the convenience of a domestic sale.
Continue to Iris Biotech GmbHSend request to US distributorPublished on 19/04/2022

Within cells, the surveillance and maintenance of protein concentration, conformation, and subcellular localization, also known as protein homeostasis, plays a crucial role for intact functioning and regulation of cell processes such as gene transcription and cell cycle control. This proteostasis is comprised of a highly interconnected set of pathways. Within eukaryotic cells, damaged, aggregated, and misfolded proteins can be cleared by proteasomes before leading to the pathogenesis of certain disorders.
To be targeted for proteasome-mediated degradation, proteins are covalently tagged with Ubiquitin (Ub) moieties. This labeling requires the sequential activity of three enzymes, namely E1 Ub-activating enzyme (E1), E2 Ub-conjugating enzyme (E2), and E3 Ub-ligase (E3). First, E1 is forming a high-energy sulfur lipid bond between itself and the ubiquitin molecule in an ATP-dependent manner resulting in an E1-Ub conjugate. The activated ubiquitin is then transferred from E1 to E2 via a trans-thioesterification reaction. At the same time, E3 binds the E2-Ub conjugate favoring the transfer of the ubiquitin molecule to the target protein. Polyubiquitinated target proteins are then recognized and degraded by the proteasome, whereas the ubiquitin molecules dissociate from the substrate and return to the cytoplasm for reutilization.
This “cell-own degradation machinery” can be employed to target disease-related proteins – a strategy that has gained tremendous attention within the last years. Targeted protein degradation (TPD) has the potential to “drug the undruggable”, thus, to modulate proteins that are difficult or impossible to target with conventional small molecules. Amongst more than 4000 identified disease-associated proteins, only around 400 proteins are successfully exploited in current therapies. Reasons for being intractable include broad active sites and shallow pockets that are difficult to bridge with small molecules or “smooth” surfaces with only few binding sites.
The concept of TPD via PROteolysis-Targeting Chimeras (PROTACs®), also known as bivalent chemical protein degraders, was developed by Crews and Deshaies groups in 2001 and has been successfully applied to multiple targets with different subcellular localization, especially in the hijacking of cancer-related kinases. Besides the above-mentioned aspect, PROTACs® afford multiple other advantages compared to traditional small molecule inhibitors. Most importantly, PROATCs® are active in a substoichiometric and catalytic manner, which allows them to function at low concentrations, thereby reducing possible toxic side effects.
PROTACs® consist of three components: one ligand with high affinity for the E3 ubiquitin ligase, another one with high affinity for the protein of interest (POI) and an appropriate cross-linker joining both ligands. This linker can also be used to increase the solubility, if needed, e.g. by incorporation of polyethylene glycol moieties. The three-component PROTAC® recruits the POI and the E2-associated E3 ligase and a ternary complex is formed. As described above, the proximity of both leads to polyubiquitination and finally to degradation through the proteasome.
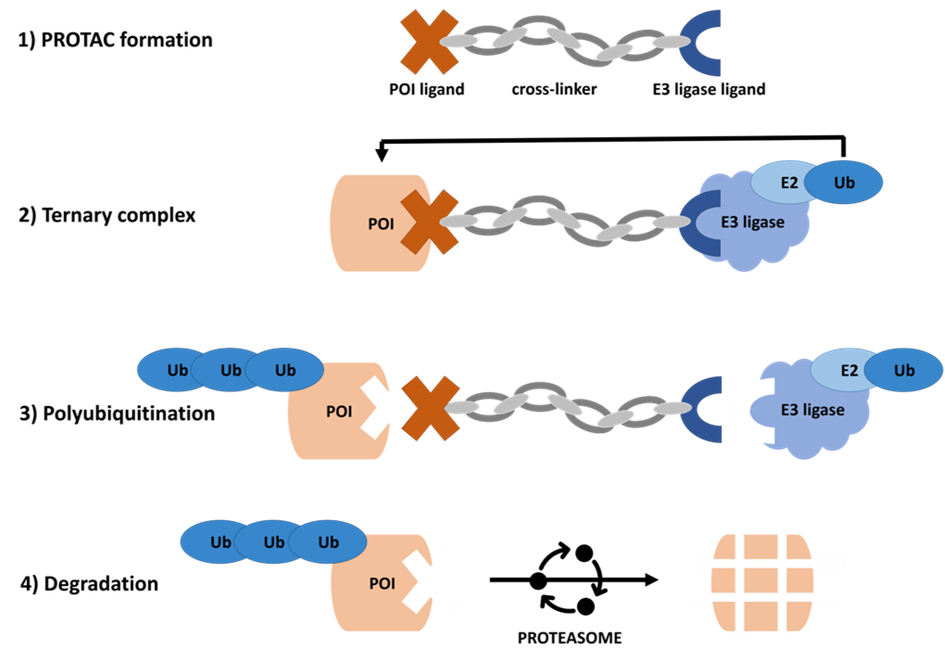
Mode of action of a PROTAC®.
To evaluate a PROTAC’s® protein degradation activity, it is incubated at a range of concentrations with selected cells expressing the POI for two to 24 h. Afterwards, a western blot is typically used to visualize and quantitate the cellular levels of the POI.
To construct and proof a suitable PROTAC®, Iris Biotech offers a variety of E3 ubiquitin ligase ligands and negative controls, e.g. Thalidomide, Pomalidomide and Lenalidomide, as well as carboxy-, click- and thiol-reactive “partial” PROTACs® (= linker-tagged E3 ubiquitin ligase ligands). Further to these predesigned building blocks, we are happy to synthesize for you many other ligand-linker combinations or “complete” PROTACs® on a custom synthesis basis.
➔ Click here to discover our portfolio
➔ Interested in advanced linker technologies? Download our brochure about Linkerology®
PROTAC® is a registered trademark of Arvinas Operations, Inc. and is used under license.
Linkerology® is a registered trademark of Iris Biotech GmbH.
References:
Targeted protein degradation: mechanisms, strategies and application; L. Zhao, J. Zhao, K. Zhong, A. Tong, D. Jia; Signal Transduction and Targeted Therapy 2022; 7(113). https://doi.org/10.1038/s41392-022-00966-4.
Protacs: chimeric molecules that target proteins to the Skp1-CullinF box complex for ubiquitination and degradation; K. M. Sakamoto, K. B. Kim, A. Kumagai, F. Mercurio, C. M. Crews, R. J. Desaies; Proc. Natl. Acad. Sci. USA 2001; 98: 8554-8559. https://doi.org/10.1073/pnas.141230798.
Recent Developments in PROTAC-Mediated Protein Degradation: From Bench to Clinic; Z. Hu, C. M. Crews; ChemBioChem 2022; 23(2). https://doi.org/10.1002/cbic.202100270.
PROTAC: An Effective Targeted Protein Degradation Strategy for Cancer Therapy; S.-M. Qi, J. Dong, Z.-Y. Xu, X.-D. Cheng, W.-D. Zhang, J.-J. Qin; Front. Pharmacol. 2021; 12: 692574. https://doi.org/10.3389/fphar.2021.692574.
Proteolysis-Targeting Chimera (PROTAC): Is the Technology Looking at the Treatment of Brain Tumors? L. L. Severini, F. Bufalieri, P. P. Infante, L. D. Marcotullio; Front. Cell Dev. Biol. 2022; 10: 854352. https://doi.org/10.3389/fcell.2022.854352.
PROTAC targeted protein degraders: the past is prologue; M. Békés, D. R. Langley, C. M. Crews; Nat. Rev. Drug Discov. 2022; 21: 181-200. https://doi.org/10.1038/s41573-021-00371-6.
Proteolysis-targeting chimera (PROTAC) for targeted protein degradation and cancer therapy; X. Li, Y. Song; J. Hematol. Oncol. Pharm. 2020; 13(50). https://doi.org/10.1186/s13045-020-00885-3.