Welcome to Iris Biotech
For better service please confirm your country and language we detected.

For better service please confirm your country and language we detected.

Thank you very much for your interest in our products. All prices listed on our website are ex-works, Germany, and may attract customs duties when imported.
You may/will be contacted by the shipping company for additional documentation that may be required by the US Customs for clearance.
We offer you the convenience of buying through a local partner, Peptide Solutions LLC who can import the shipment as well as prepay the customs duties and brokerage on your behalf and provide the convenience of a domestic sale.
Continue to Iris Biotech GmbHSend request to US distributorPublished on 23/03/2021
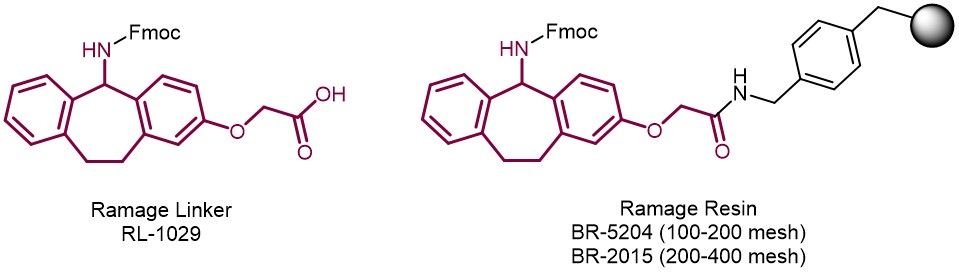
Iris Bioech offers the Ramage-Linker (RL-1029) in high quality available in bulk quantities. The basis for the Ramage linker is a dibenzocyclohepta-1,3-dienyl system with an additional oxygen substituent which serves on one hand for further stabilization, and on the other hand as site of appendage to the polystyrene resin. For your convenience, we also offer Ramage-linker polystyrene resins in different mesh sizes (BR-5204, BR-2015).
The Ramage linker represents an alternative to the commonly used Rinklinker for the synthesis of C-terminal peptide amides. The three-circular structure of the Ramage linker prevents fragmentation of the linker during cleavage from the resin followed by back alkylation. This is a drawback commonly observed during peptide synthesis with the Rink-amide linker. Consequently, the Ramage linker delivers peptides in higher purity with less impurities compared to peptides produced with the Rink-amide linker.
After Fmoc removal, standard SPPS can be carried out with conventional Fmoc/tBu protocols. Peptide cleavage from the resin can be achieved by treatment with only 3% TFA in DCM to yield the fully protected peptide, or with 50% TFA in DCM or 95% aqueous TFA to remove any side chain protecting groups.
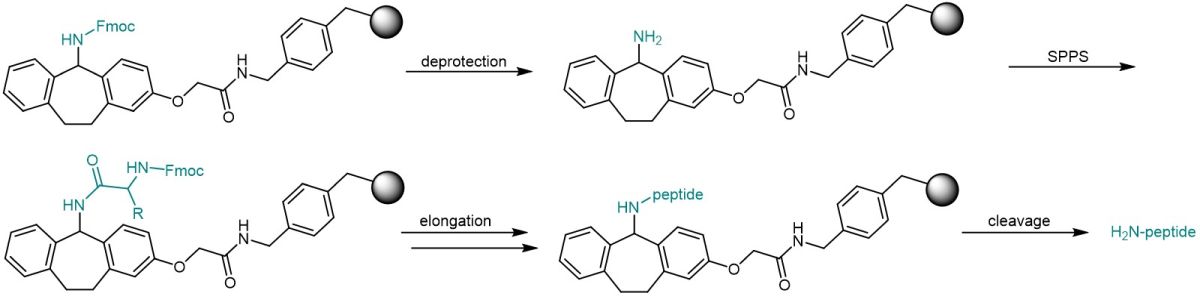
Standard solid-phase peptide synthesis using the Ramage resin.
Besides C-terminal amides, the Ramage linker can also be used for the synthesis of C-terminal peptide alcohols. Their diverse biological activities and clinical importance are described in various publications. One prominent example is Octreotide, a metabolically stable analog of somatostatin which contains threoninol at the C-terminus. Starting from the Ramage linker or resin, the deprotected primary amine can be converted to a hydroxy moiety and further to the corresponding chloride, which can then be loaded with Fmoc-amino alcohols. Analogous to the preparation of peptide amides, the synthesized sequence can be detached from the linker and fully deprotected by addition of 95% aqueous TFA, while treatment with 20% hexafluoroisopropanol in dichloromethane is reported to render a fully protected peptide alcohol that can be further modified at the C-terminus.
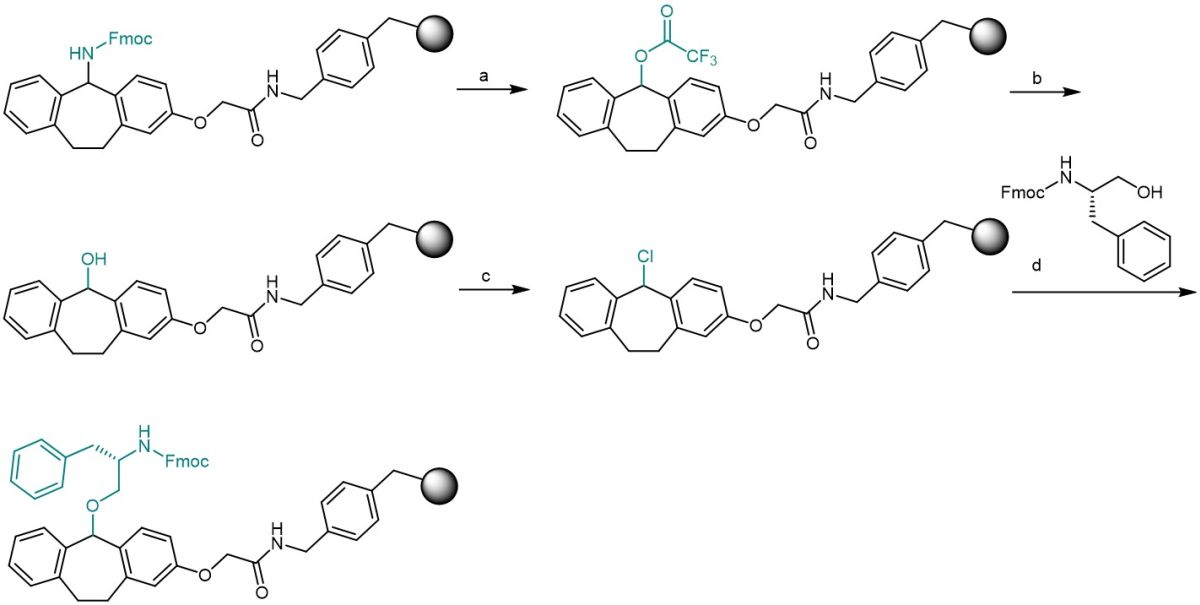
Exemplary synthesis of Fmoc-phenylalaninol linked to the Ramage Resin: a) 1 M solution of trifluoroacetic anhydride in 2,6-lutidine for 30 min. b) 0.4 M K2SO4 in dioxane-water 7:1 for 16 h. c) PPh3 C2Cl6 (3 eq.) in THF room temperature for 12 h. d) Fmoc-amino alcohol (5 eq.), 2,6 lutidine (5.5 eq.), microwave at 90 °C in DMF for 15 min.
References:
Design of a versatile linker for solid phase peptide synthesis: Synthesis of C-terminal primary/seconary amides and hydrazides; R. Ramage, S. L. Irving and C. McInnes; Tetrahedron Lett 1993; 34: 6599-6602. https://doi.org/10.1016/0040-4039(93)88115-y
Functionalized Resins for the Synthesis of Peptide Alcohols; F. J. Ferrer Gago, L. Q. Koh, D. P. Lane; Chem. Eur. J. 2020; 26(2): 379-383. https://doi.org/10.1002/chem.201903965.