Welcome to Iris Biotech
For better service please confirm your country and language we detected.

For better service please confirm your country and language we detected.

Thank you very much for your interest in our products. All prices listed on our website are ex-works, Germany, and may attract customs duties when imported.
You may/will be contacted by the shipping company for additional documentation that may be required by the US Customs for clearance.
We offer you the convenience of buying through a local partner, Peptide Solutions LLC who can import the shipment as well as prepay the customs duties and brokerage on your behalf and provide the convenience of a domestic sale.
Continue to Iris Biotech GmbHSend request to US distributorChemical name: (S)-Trifluoromethylallylglycine // Synonyms: H-alpha-Tfm-AllGly-OH
Starting at $812.50
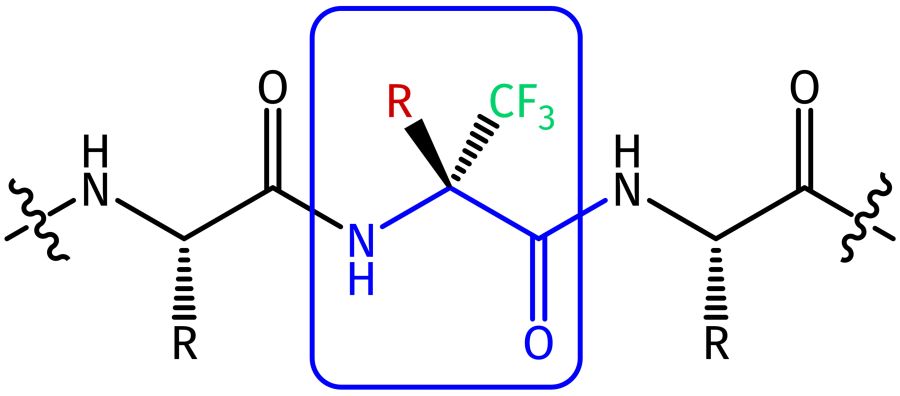
Building block which can be used for the direct implementation of a trifluoromethylated amino acid (Tfm-Aaa) at the N-terminus of a peptide. The trifluoromethyl group reduces the nucleophilicity of the amino function to a level that no further protection (e.g. by Boc, Fmoc or Z) is necessary.
For the introduction of this Tfm amino acid within the sequence the design of a dipeptide building block is required (Fmoc-Aaa-Tfm-Aaa), which can then be incorporated in a peptide sequence by standard SPPS methodologies. Please consult with our custom synthesis division.
Fluorine in Peptide Design and Protein Engineering; C. Jäckel and B. Koksch; Eur J. Org. Chem. 2005; 21: 4483-4503. https://doi.org/10.1002/ejoc.200500205
Conformational properties of peptides incorporating a fluorinated pseudoproline residue; G. Chaume, D. Feytens, G. Chassaing, S. Lavielle, T. Brigaud, E. Miclet; New J. Chem. 2013; 37: 1336-1342. https://doi.org/10.1039/C3NJ41084F
Impact of fluorination on proteolytic stability of peptides in human blood plasma; V. Asante, J. Mortier, H. Schlüter, B. Koksch; Bioorg. Med. Chem. 2013; 21: 3542-3546. https://doi.org/10.1016/j.bmc.2013.03.051.
Fluorinated Proteins: From Design and Synthesis to Structure and Stability; E. N. G. Marsh; Acc. Chem. Res. 2014; 47: 2878-2886. https://doi.org/10.1021/ar500125m
How Cα-Fluoroalkyl Amino Acids and Peptides Interact with Enzymes: Studies Concerning the Influence on Proteolytic Stability, Enzymatic Resolution and Peptide Coupling; R. Smits, B. Koksch; Current Topics in Medicinal Chemistry 2006; 6: 1483-1498. https://doi.org/10.2174/156802606777951055
Approaches to Obtaining Fluorinated α-Amino Acids; J. Moschner, V. Stulberg, R. Fernandes, S. Huhmann, J. Leppkes, B. Koksch; Chem. Rev. 2019; 119: 10718-10801. https://doi.org/10.1021/acs.chemrev.9b00024
Applications of fluorine-containing amino acids for drug design; H. Mei, J. Han, K. D. Klika, K. Izawa, T. Sato, N. A. Meanwell, V. A. Soloshonok; Eur. J. Med. Chem. 2020; 186: 111826. https://doi.org/10.1016/j.ejmech.2019.111826
Fluorinated amino acids: compatibility with native protein structures and effects on protein-protein interactions; M. Salwiczek, E. K. Nyakatura, U. I. M. Gerling, S. Ye, B. Koksch; Chem. Soc. Rev. 2012; 41: 2135-2171. https://doi.org/10.1039/C1CS15241F
Substitution Effect of the Trifluoromethyl Group on the Bioactivity in Medicinal Chemistry: Statistical Analysis and Energy Calculations; A. Abula, Z. Xu, Z. Zhu, C. Peng, Z. Chen, W. Zhu, H. A. Aisa; J. Chem. Inf. Model 2020; https://doi.org/10.1021/acs.jcim.0c00898
Synthesis of an MIF-1 analogue containing enantiopure (S)-alpha-trifluoromethyl-proline and biological evaluation on nociception; I. Jlalia, N. Lensen, G. Chaume, E. Dzhambazova, L. Astasidi, R. Hadjiolova, A. Bocheva, T. Brigaud; Eur J Med Chem 2013; 62: 122-9. https://doi.org/10.1016/j.ejmech.2012.12.041
Do you need larger quantities for your development or production?
Please send me more information about