Welcome to Iris Biotech
For better service please confirm your country and language we detected.

For better service please confirm your country and language we detected.

Thank you very much for your interest in our products. All prices listed on our website are ex-works, Germany, and may attract customs duties when imported.
You may/will be contacted by the shipping company for additional documentation that may be required by the US Customs for clearance.
We offer you the convenience of buying through a local partner, Peptide Solutions LLC who can import the shipment as well as prepay the customs duties and brokerage on your behalf and provide the convenience of a domestic sale.
Continue to Iris Biotech GmbHSend request to US distributorChemical name: N-epsilon-carboxymethyl-[D4]-L-Lysine(S)-2-amino-6-(carboxymethylamino)-4,4,5,5-tetradeuterohexanoic acid // Synonyms: H-L-Lys(Cm)-OH-d4, H-Lys(Cm)-OH-d4, N-epsilon-carboxymethyl-[4,4,5,5-D4]-L-lysine
Starting at $448.50
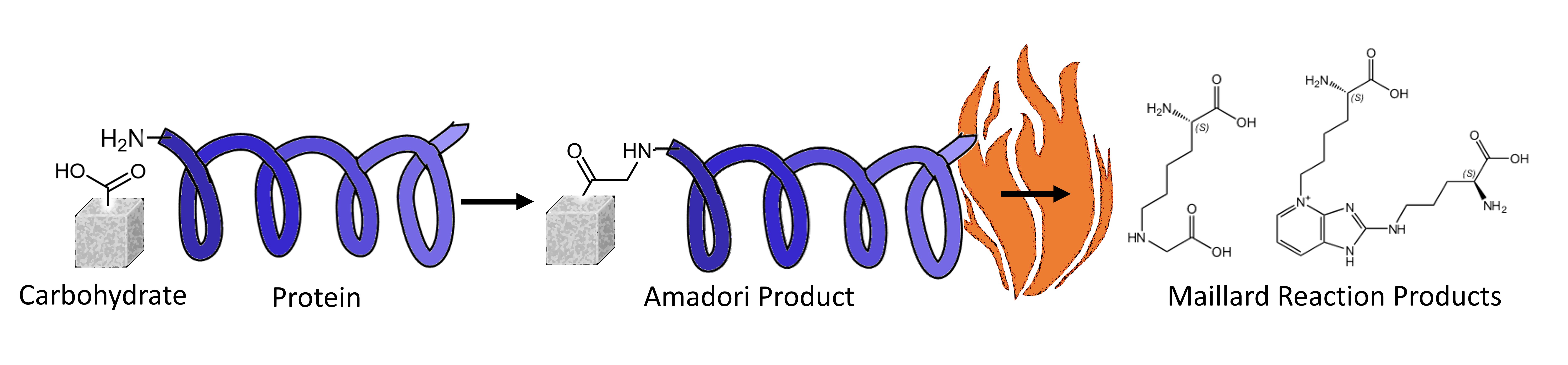
Maillard Reaction Products (MRPs) are indicators for the heat treatment of food, as well as markers of nutritional quality. They also appear naturally in the human body as so-called Advanced Glycation End Products (AGEs). As such, they are indicators for body health and disease processes, including inflammation, diabetes, cancer and ageing. Consequently, AGEs gained broad attention in cosmetics, biochemistry, food, and pharmaceutical applications.
This material is supplied as a salt containing varying contents of the acid counterion. The net content of each batch is specified in the respective certificate of analysis.
Determination of N ε-carboxymethyllysine in heated milk products by immunochemical methods; A. Tauer, K. Hasenkopf, T. Kislinger, I. Frey and M. Pischetsrieder; European Food Research and Technology 1999; 209: 72-76. https://doi.org/10.1007/s002170050460
N epsilon-(carboxymethyl)lysine is a dominant advanced glycation end product (AGE) antigen in tissue proteins; S. Reddy, J. Bichler, K. J. Wells-Knecht, S. R. Thorpe and J. W. Baynes; Biochemistry 1995; 34: 10872-8. https://doi.org/10.1021/bi00034a021
Mechanism of protein modification by glyoxal and glycolaldehyde, reactive intermediates of the Maillard reaction; M. A. Glomb and V. M. Monnier; J Biol Chem 1995; 270: 10017-26. https://doi.org/10.1074/jbc.270.17.10017
Mechanism of the degradation of non-enzymatically glycated proteins under physiological conditions. Studies with the model fructosamine, N epsilon-(1-deoxy-D-fructos-1-yl)hippuryl-lysine; P. R. Smith and P. J. Thornalley; Eur J Biochem 1992; 210: 729-39. https://doi.org/10.1111/j.1432-1033.1992.tb17474.x
Oxidative degradation of glucose adducts to protein. Formation of 3-(N epsilon-lysino)-lactic acid from model compounds and glycated proteins; M. U. Ahmed, J. A. Dunn, M. D. Walla, S. R. Thorpe and J. W. Baynes; J Biol Chem 1988; 263: 8816-21.
Do you need larger quantities for your development or production?
Please send me more information about