Welcome to Iris Biotech
For better service please confirm your country and language we detected.

For better service please confirm your country and language we detected.

Thank you very much for your interest in our products. All prices listed on our website are ex-works, Germany, and may attract customs duties when imported.
You may/will be contacted by the shipping company for additional documentation that may be required by the US Customs for clearance.
We offer you the convenience of buying through a local partner, Peptide Solutions LLC who can import the shipment as well as prepay the customs duties and brokerage on your behalf and provide the convenience of a domestic sale.
Continue to Iris Biotech GmbHSend request to US distributorPublished on 14.09.2017
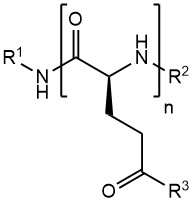
Their highly charged nature makes PGAs an ideal component for the coating/formation of nanoparticles. Furthermore, the multitude of gamma-carboxyl groups can serve as attachment points for different kinds of molecules. One well-known application is the formation of PGA-API conjugates, such as PGA-Paclitaxel. In addition to APIs, the PGA polymer can also be functionalized with targeting moieties, imaging agents or solubilizing moieties.
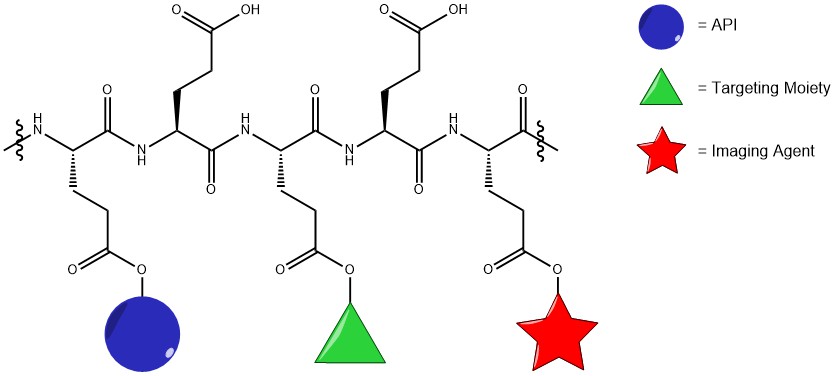
Ringsdorf's model for a targetable polymer-drug conjugate (Ringsdorf, J. Polym. Sci. Polym. Symp. 1975).
Our PGAs are available with lengths from 20 to 800 glutamic acid units, as well as with a multitude of different functional groups and other modifications:
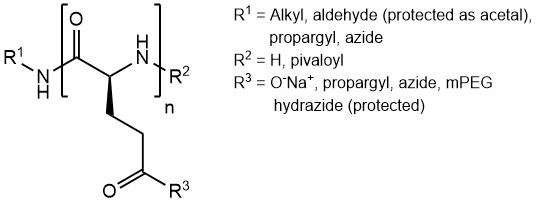
Modifications on the termini (R¹ and R²) include alkyl residues, alkyne groups and protected aldehydes. Among the available side-chain modifications (R³) are alkynes, azides, protected hydrazides and PEG-chains.
→ Find all of our PGA derivatives (more than 80 different compounds) in the Poly-Glutamic Acids section of our webshop.
→ Be sure to also have a look at our large and diverse selection of Polymeric Carriers, including mono- and polydisperse PEGs, various polyamino acids, as well as polyoxazolines.