Welcome to Iris Biotech
For better service please confirm your country and language we detected.

For better service please confirm your country and language we detected.

Thank you very much for your interest in our products. All prices listed on our website are ex-works, Germany, and may attract customs duties when imported.
You may/will be contacted by the shipping company for additional documentation that may be required by the US Customs for clearance.
We offer you the convenience of buying through a local partner, Peptide Solutions LLC who can import the shipment as well as prepay the customs duties and brokerage on your behalf and provide the convenience of a domestic sale.
Continue to Iris Biotech GmbHSend request to US distributorPublished on 29.09.2020
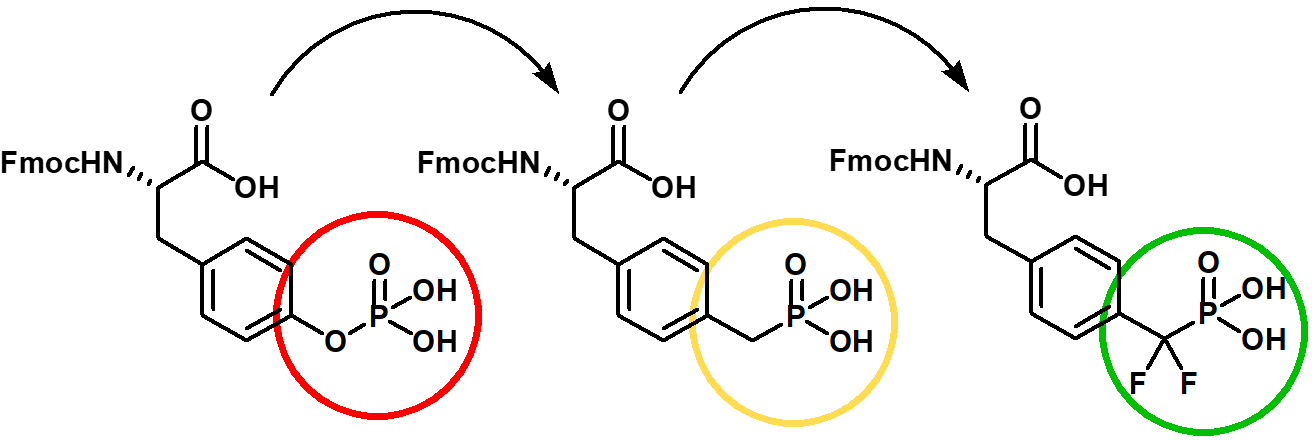
The preparation of synthetic phosphorylated peptides is of significant interest for research. Among posttranslational modifications that occur in organisms, phosphorylation of serine, threonine and tyrosine is counted among the most important activating ones. Nevertheless, from a chemical point of view, those phosphoesters are highly hydrolysis labile. Iris Biotech’s portfolio already includes phosphono-amino acid derivatives which serve as hydrolysis-stable mimics of pSer, pThr and pTyr, termed Pma (Ser), Pmab (Thr) and Pmp (Tyr).
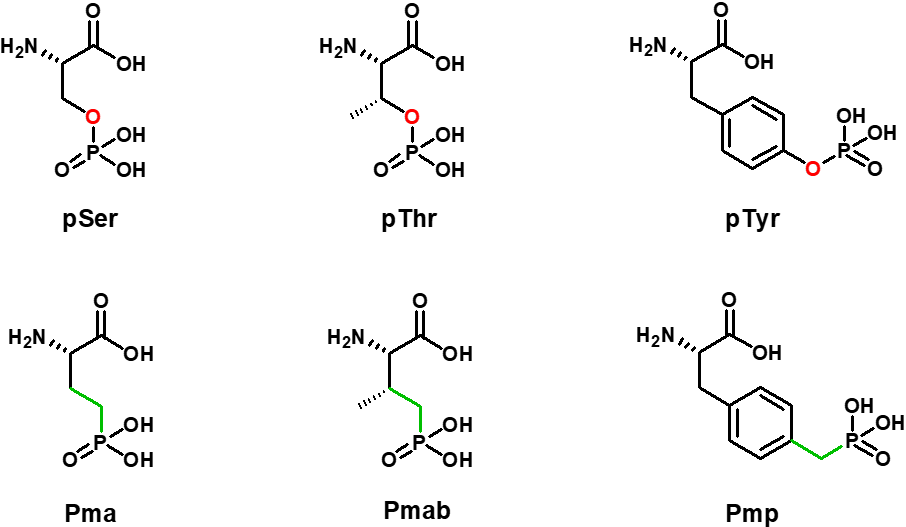
Phosphoester Serine, Threonine and Tyrosine and its hydrolysis stable phosphono derivatives.
The stability towards hydrolysis that characterizes the phosphono-derivatives has an additional benefit, which is that cellular phosphatases are unable to remove the phosphate group mimic. Consequently, peptides or semi-synthetic proteins that include Pma, Pmab or Pmp are valuable tools for cell-based experiments.
As further innovative improvement, Iris Biotech now expanded the range of hydrolysis-stable mimics. Our new phosphono tyrosine derivative is difluorinated which renders the phosphonic acid more acidic, thus becoming a better mimic of phosphotyrosine than Pmp while keeping the hydrolytic stability. If you are interested in the difluoro phosphono derivatives of serine and threonine, please get in contact – we will provide those based on a custom synthesis inquiry.

Fmoc-protected phosphorylated tyrosine and the hydrolysis stable Pmp and difluoro-Pmp derivative.
All derivatives are suitably protected for use in peptide synthesis by Fmoc strategy. The tert-butyl protecting groups prevent side reactions and can be removed during final deprotection of the peptide.
References: