Welcome to Iris Biotech
For better service please confirm your country and language we detected.

For better service please confirm your country and language we detected.

Thank you very much for your interest in our products. All prices listed on our website are ex-works, Germany, and may attract customs duties when imported.
You may/will be contacted by the shipping company for additional documentation that may be required by the US Customs for clearance.
We offer you the convenience of buying through a local partner, Peptide Solutions LLC who can import the shipment as well as prepay the customs duties and brokerage on your behalf and provide the convenience of a domestic sale.
Continue to Iris Biotech GmbHSend request to US distributorPublished on 23/03/2022

The choice of a linker for selective and site-specific control of payload release remains a major goal during drug delivery. The cleavage mechanism of disulfide-based linkers is based on the specific intracellular release of the active payload upon glutathione (GSH) reduction.
As the overall red/ox potential in the human blood is oxidative, disulfide linkages remain intact during circulation. However, intracellularly, and notably inside tumor mass and cancer cells, a reductive potential is prevailing. This discrepancy in GSH concentration allows to develop GSH-responsive materials and to revert the disulfide bond formation selectively in pathological tissues and thus release the active payload.
Iris Biotech presents a selection of pyridyl-disulfide-nitrophenylcarbonate linkers (see also related products).
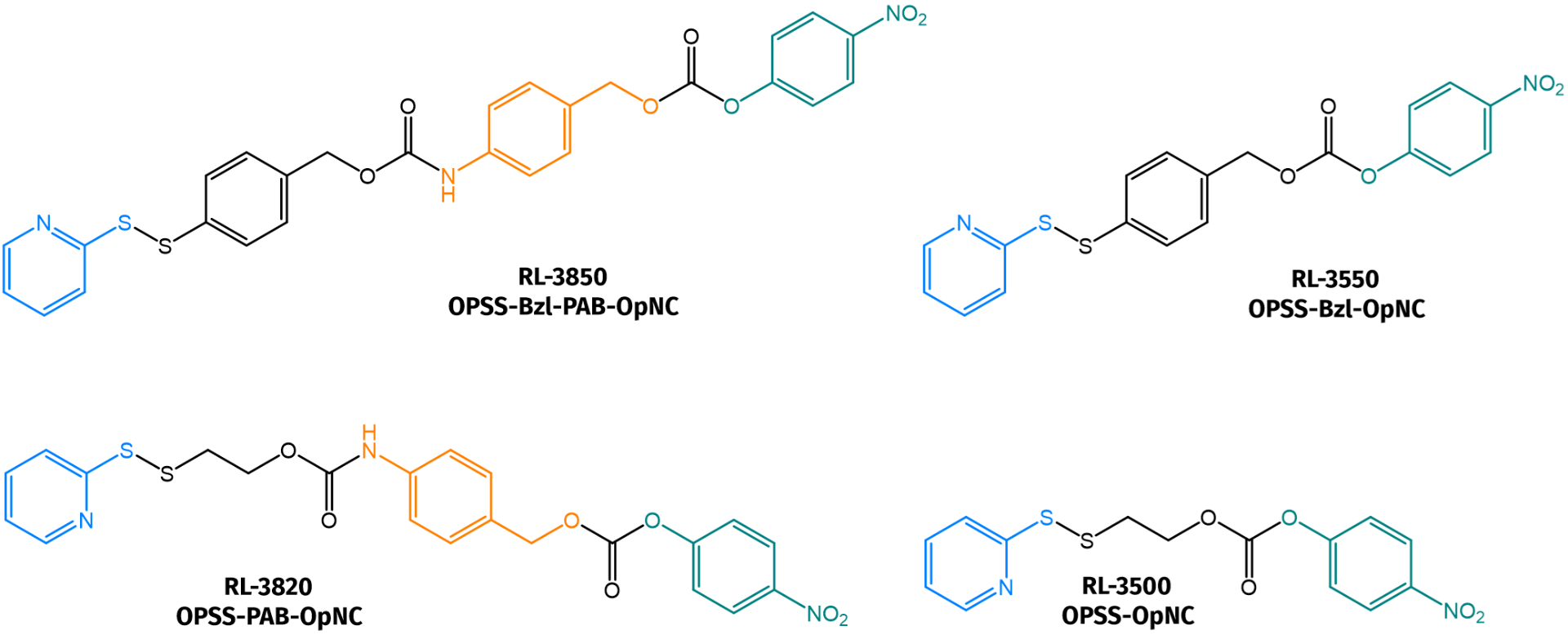
Selection of pyridyl-disulfide-nitrophenylcarbonate linkers available at Iris Biotech.
The pyridyl disulfide moiety can undergo a disulfide exchange reaction with sulfhydryl groups, e.g. from a biomolecule, to form new disulfide bonds. Thereby, pyridine-2-thione is released, which can be followed spectrophotometrically (λmax = 343 nm) to measure the progress of the reaction. This reaction works over a broad pH range and is also suitable for physiological pH.
The p-nitrophenylcarbonate activating group reacts preferably with amines or other nucleophiles and thus allows further conjugation, e.g. towards the desired drug molecule.
Finally, the p-aminobenzyl moiety acts as self-immolative linkage (SIL) and disintegrates upon a certain trigger, e.g. disulfide reduction, to release the conjugated payload in a traceless manner.
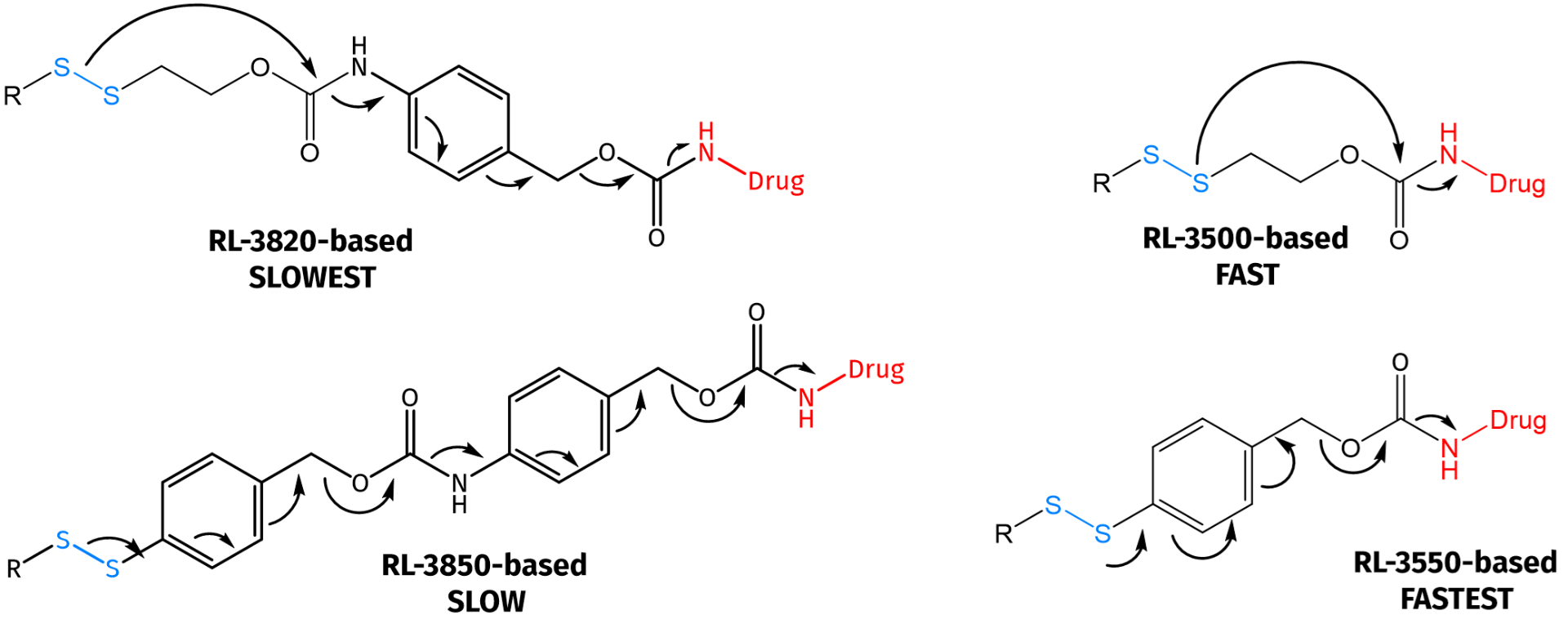
Tunable kinetics of self-immolative drug release.
SILs can be mainly classified into two groups depending on the self-immolative fragmentation process either based on the quinone methide cascade (e.g. 1,6-elimination) or an intramolecular cyclization reaction. In addition, the linker’s chemical composition (e.g. disulfide ethoxycarbonyl (SSE) vs. disulfide benzyloxycarbonyl (SSB)) results in chemically tunable kinetics of the self-immolative cleavage due to different response rate towards GSH, showing higher rates for SSB-based DSILs compared to SSE-based ones. Thus, the choice of the linker allows for fine-tuning of the cleavage speed and payload release.
References:
Disulfide-Based Self-Immolative Linkers and Functional Bioconjugates for Biological Applications; Z. Deng, J. Hu and S. Liu; Macromol Rapid Commun 2020; 41: e1900531. https://doi.org/10.1002/marc.201900531