Welcome to Iris Biotech
For better service please confirm your country and language we detected.

For better service please confirm your country and language we detected.

Thank you very much for your interest in our products. All prices listed on our website are ex-works, Germany, and may attract customs duties when imported.
You may/will be contacted by the shipping company for additional documentation that may be required by the US Customs for clearance.
We offer you the convenience of buying through a local partner, Peptide Solutions LLC who can import the shipment as well as prepay the customs duties and brokerage on your behalf and provide the convenience of a domestic sale.
Continue to Iris Biotech GmbHSend request to US distributorPublished on 20/01/2023

Nowadays we know that alkynes and azides can undergo a Cu(I)-catalyzed azide-alkyne 1,3-dipolar cycloaddition (CuAAC) to afford the 1,4-disubstituted triazoles. This type of chemical transformation was dubbed “Click Chemistry” and awarded with the 2022 Nobel Prize for Chemistry.
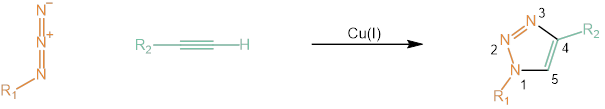
Schematic illustration of the click reaction between an azide and an alkyne.
The reactivity of an alkyne strongly depends on the electronic and steric characteristics of neighbouring substituents as well as structural strain. In comparison to linear acyclic alkynes, strained cyclic alkynes show remarkably high reactivity without addition of a catalyst, e.g. copper, as typically used for click chemistry. Thus, the metal-free strain-promoted azide-alkyne cycloaddition (SPAAC) is a valuable tool especially for future in vivo applications.
Many SPAAC reagents are hydrophobic, show slow conversion, are incompatible with biomolecules or are too unstable. Herein, we present a novel SPAAC alkyne reagent, which overcomes the aforementioned drawbacks.
The CliCr® reagent meets all demands required in the life science industry by its innovative TMTHSI moiety. The 7-membered ring can be conveniently functionalized with a variety of linkers, e.g. via acylation, sulfonylation, N-alkylation, or carbamoylation.
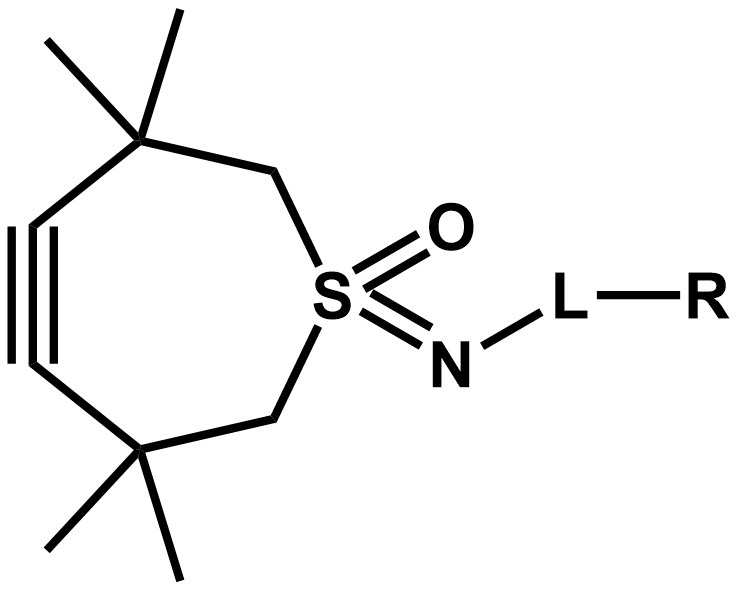
Chemical structure of the CliCr® base compound and its derivatization possibilities.
The versatility of CliCr-reagents to conjugating other moieties has been illustrated by the reaction of TMTHSI-derivatives with small molecules, exemplified by the click-reaction with a dye and a folic acid derivative, as well as peptides, proteins (including ADC), nucleic acid biologics and nanoparticles. Besides this, a plethora of additional important applications is envisioned, such as surface plasmon resonance (SPR) applications and conjugation of chelator moieties for radioactive isotope incorporation in theragnostic applications.
In comparison to other strained alkynes, CliCr® creates faster, more hydrophilic bioconjugates with superior profiles and overall more attractive costs of goods.
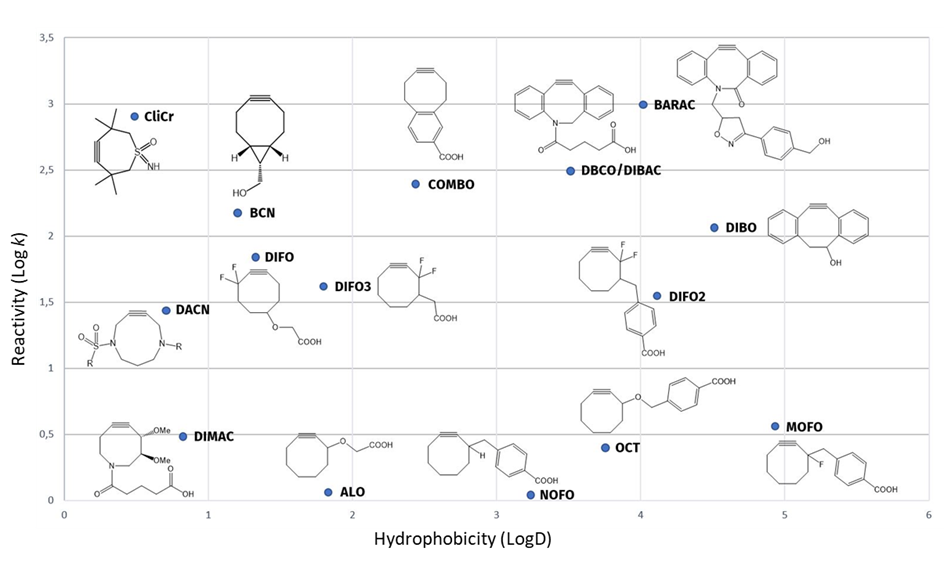
Reactivity and hydrophilicity of CliCr® compared to other strained alkynes.
CliCr® is shown to be > 5 times more reactive than BCN. The reaction progress of 5 mM CliCr® or BCN-OH, respectively, with 1.3 eq. of benzylazide in CDCl3 at room temperature was monitored by MS. The reaction conversion was measured based on the increase of the triazole signals (see https://doi.org/10.1039/d0sc03477k).
Within our portfolio, we offer both CliCr® as a base compound and a selection of CliCr® derivatives. The CliCr® derivatives can be functionalized with a moiety of your own preference and subsequently clicked to azide compounds with high efficiency and excellent stability. For other derivatives, large scale production, or GMP grade, please get in contact!
CliCr® is provided under an intellectual property license from Cristal Therapeutics. The trademark CliCr® is the property of Cristal Therapeutics. For information on acquiring a license to this technology for purpose other than as described above, please contact Cristal Therapeutics, Oxfordlaan 55, 6229 EV Maastricht, The Netherlands info@cristaltherapeutics.com.
References:
TMTHSI, a superior 7-membered ring alkyne containing reagent for strain-promoted azide-alkyne cycloaddition reactions; J. Weterings, C. J. F. Rijcken, H. Veldhuis, T. Meulemans, D. Hadavi, M. Timmers, M. Honing, H. Ippel, R. M. J. Liskamp; Chem. Sci. 2020; 11: 9011-9016. https://doi.org/10.1039/d0sc03477k
https://cristaltherapeutics.com/technology/clicr-copper-free-click-chemistry-reagent/