Welcome to Iris Biotech
For better service please confirm your country and language we detected.

For better service please confirm your country and language we detected.

Thank you very much for your interest in our products. All prices listed on our website are ex-works, Germany, and may attract customs duties when imported.
You may/will be contacted by the shipping company for additional documentation that may be required by the US Customs for clearance.
We offer you the convenience of buying through a local partner, Peptide Solutions LLC who can import the shipment as well as prepay the customs duties and brokerage on your behalf and provide the convenience of a domestic sale.
Continue to Iris Biotech GmbHSend request to US distributorPublished on 18/05/2021
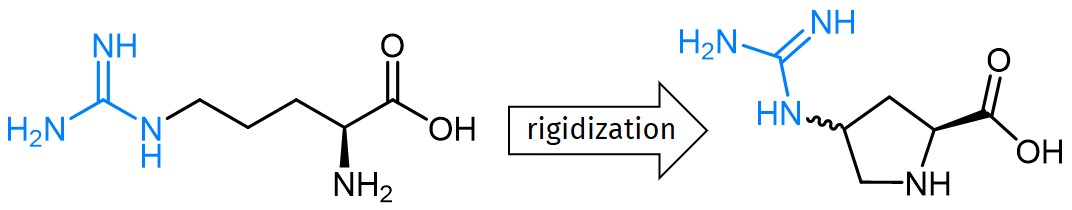
The site-directed replacement of one amino acid within a peptide sequence or protein allows to determine the role of this individual element for peptide/protein function and to investigate its effect on receptor interactions. Especially the incorporation of structurally closely related, but conformationally different derivatives provides a high level of functional resolution. Finally, such structure-activity data allow the fine-tuning e.g. of peptide drugs for optimized receptor binding and maximized efficiency and activity.
For the methodical study of Arginine containing peptides, our portfolio includes two guandinylated Proline derivatives, trans- and cis-ProGU. The strong basic guanidino group of Arginine, which is almost always protonated at physiological pH, plays a pivotal role in molecular recognition processes. It is crucial for hydrogen bonding as well as ionic interactions with binding partners. In contrast to the rather flexible guanidino group of Arginine, trans-ProGU and cis-ProGU represent constrained mimics. In both Proline derivatives, the guanidinium moiety is directly attached to the gamma carbon. Thus, the positively charged moiety is rather localized, which allows to study the topographic importance of the guanidinium group for receptor interaction and peptide activity.
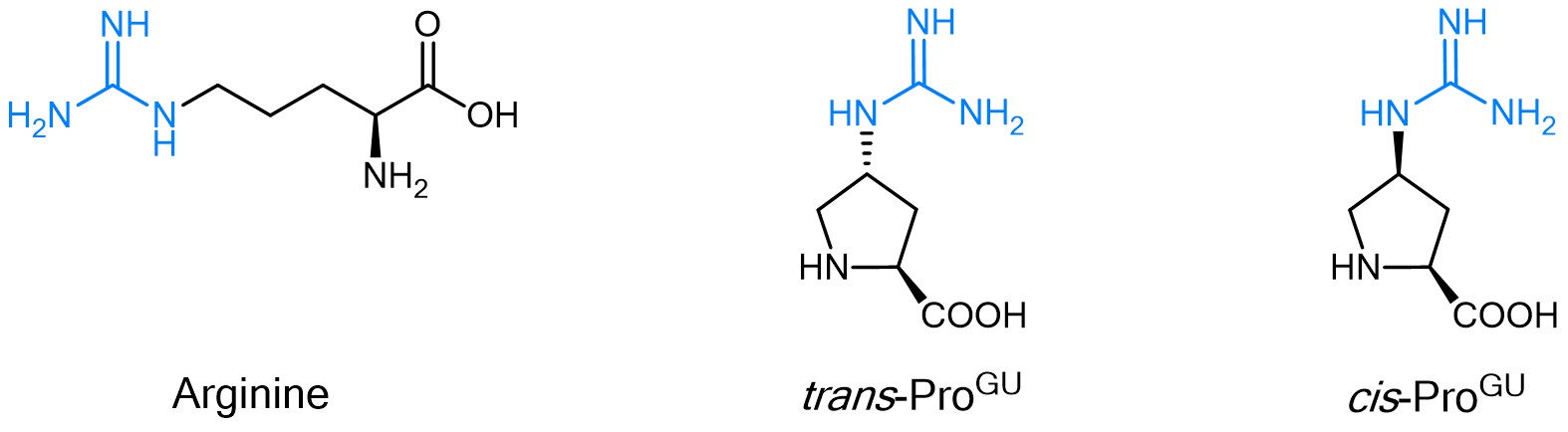
The constrained Arginine mimics trans-ProGU and cis-ProGU.
As an example, the introduction of a guanidinylated Proline in Arg-rich peptides resulted in 2.5- to 15-fold higher cellular uptake and approximately 100-fold more efficient pDNA transfection efficiencies compared to R9 peptides. Within our portfolio, we offer two guanidinylated Proline derivatives, which are listed at the end of this article under “related products”. Those Fmoc-protected guanidino-Pbf derivatives can easily be introduced as building blocks during peptide synthesis.
Interested in other Arginine derivatives? Check-out our webshop or inquire for a custom synthesis.
References: