Welcome to Iris Biotech
For better service please confirm your country and language we detected.

For better service please confirm your country and language we detected.

Thank you very much for your interest in our products. All prices listed on our website are ex-works, Germany, and may attract customs duties when imported.
You may/will be contacted by the shipping company for additional documentation that may be required by the US Customs for clearance.
We offer you the convenience of buying through a local partner, Peptide Solutions LLC who can import the shipment as well as prepay the customs duties and brokerage on your behalf and provide the convenience of a domestic sale.
Continue to Iris Biotech GmbHSend request to US distributorPublished on 11/01/2021
Native chemical ligation (NCL) remains the method of choice for joining two unprotected peptide fragments. In the context of Fmoc solid-phase peptide synthesis (SPPS), the crucial step is the efficient synthesis of C-terminal peptide-thioesters, as the methods established for Boc-SPPS have only limited utility due to the lability of the thioester linkage under basic Fmoc-removal conditions.
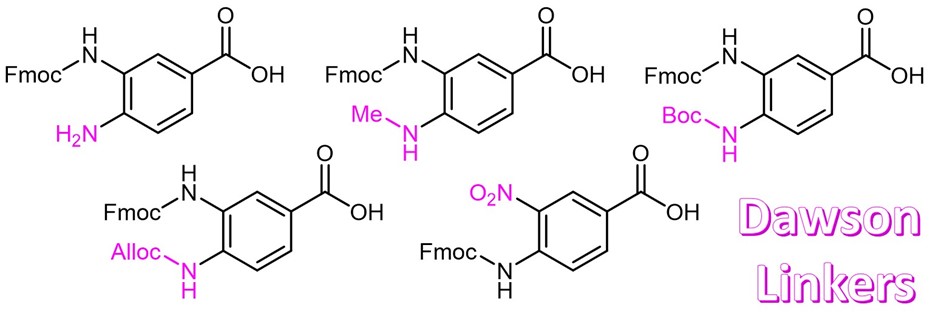
The most common strategy for the preparation of a peptide thioester surrogate by Fmoc chemistry is based on the use of a 3,4-diaminobenzoic acid (Dbz) derivative (= Dawson linker). Iris Biotech offers this linker (FAA3165) and various derivatives with a Fmoc protecting group. After Fmoc removal, the resin is being acylated with the first amino acid and peptide elongation can be performed. After successful chain assembly, the resin is activated by reaction of the remaining Dbz amine with para-nitrophenyl chloroformate, followed by rearrangement and cyclization to N-acyl-benzimidazolinone (Nbz) in basic media. Finally, trifluoroacetic acid cleaves the fully deprotected peptide benzimidazolinone which can then be converted to a thioester upon reaction with an aryl thiol.
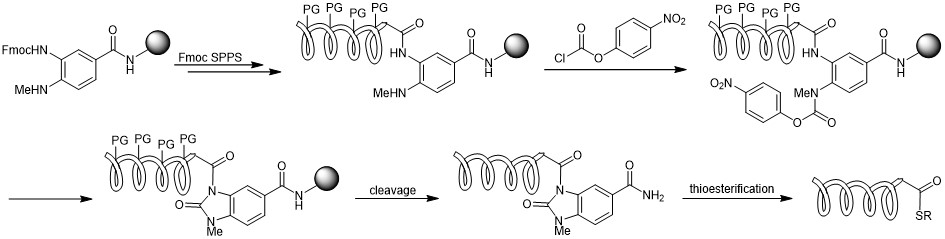
Scheme for the thioester generation by using Me-Dbz during Fmoc SPPS.
The main difficulty during the formation of those thioester surrogates remains the overacylation of the deactivated ortho-amino group of the Dbz linker. Thus, its protection is required to avoid the accumulation of branched and acetylated side products and to better control chain extension.
The methylation of the Fmoc-Dbz amine in position 4 makes it less reactive and suppresses the formation of side products on the para-amino moiety. Thus, MeDbz (FAA3166) allows the rather selective elongation of the peptide chain on the amino function in position 3 compared to the first-generation Dbz linker FAA3165.
Another derivative (FAA3168) is based on the allyloxy-carbonyl (Alloc) protection of the Dbz amine in position 4. After successful synthesis, the Alloc group can quantitatively be removed under mild conditions, and finally the thioester can be formed.
FAA3167 makes use of the tert-butyloxycarbonyl (Boc) group as efficient and acid-labile protecting group for the deactivated Dbz amine. Unlike Alloc, the Boc group can easily be removed by TFA during the global deprotection of the peptide from the resin without the need for an additional step.
Especially for microwave-assisted peptide synthesis, the 4-amino-3-nitrobenzoic acid linker (FAA3169) was developed. After successful chain assembly, Dbz(NO2) can easily be converted into the Dbz linker by on-resin reduction with SnCl2. Even though epimerization of the C-terminal amino acid limits the use of the nitro derivative to the synthesis of peptide-Gly-thioesters, it suppresses the formation of side-products which are otherwise observed during microwave-assisted synthesis when using Dbz or Dbz(Alloc).
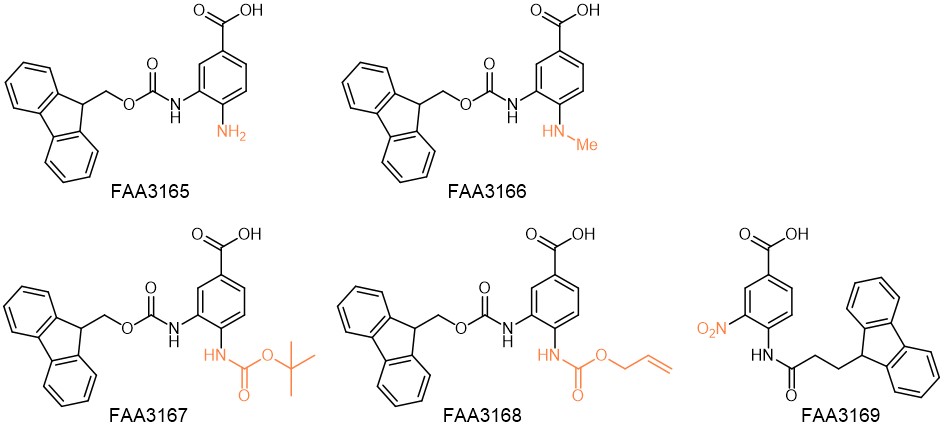
Dawson linker derivatives provided by Iris Biotech.
Interested in more information on Native Chemical Ligation? -> Please feel free to download our new brochure!
References: