Welcome to Iris Biotech
For better service please confirm your country and language we detected.

For better service please confirm your country and language we detected.

Thank you very much for your interest in our products. All prices listed on our website are ex-works, Germany, and may attract customs duties when imported.
You may/will be contacted by the shipping company for additional documentation that may be required by the US Customs for clearance.
We offer you the convenience of buying through a local partner, Peptide Solutions LLC who can import the shipment as well as prepay the customs duties and brokerage on your behalf and provide the convenience of a domestic sale.
Continue to Iris Biotech GmbHSend request to US distributorPublished on 28/06/2022

Native Chemical Ligation (NCL) is a powerful protocol that is frequently used in the synthesis of complex peptide sequences. The NCL technique also grants access to small fully synthetic or large semi-synthetic proteins. One of the two peptide fragments required for NCL has to be converted to a C-terminal thioester. This step necessitates either special synthetic conditions for SPPS that frequently result in racemization and/or low yields, or additional modifications in solution phase that lead to more tedious isolation and purification steps.
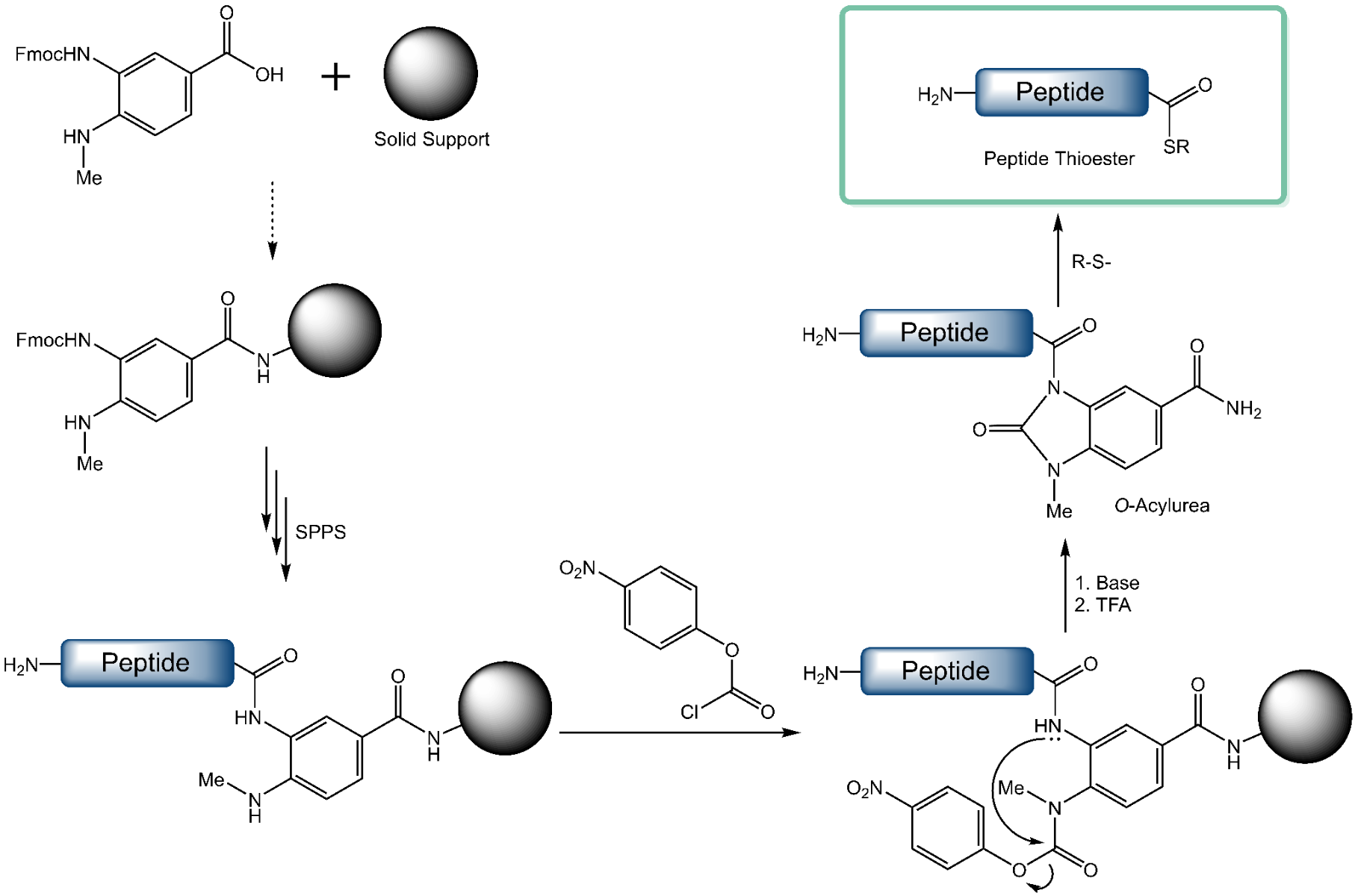
A practical and synthetically more accessible thioester surrogate in the form of an O-acylurea peptide can be prepared by utilizing the innovative Dawson-Linker (Fmoc-MeDbz-OH). After successful chain assembly on resin, the linker is activated by reaction of its secondary amine with para-nitrophenyl chloroformate. The N-methyl group of the 4-amino functionality of Fmoc-MeDbz-OH hereby suppresses potential side reactions occurring at this position. This step is followed by a base-catalyzed rearrangement and cyclization to N-acyl-N’-methyl-benzimidazolinone (MeNbz). Cleavage from the resin with trifluoroacetic acid affords the O‑acylurea peptide which may then either be converted to a C-terminal thioester by reaction with an aryl thiol or may be used directly for Native Chemical Ligation.
Iris Biotech offers the classical Dawson linker (FAA3165) as well as various of its derivatives (see “Related Products” below). As previously mentioned, the main difficulty during the formation of the O-acylurea peptides is the overacylation of the deactivated ortho-amino group of the Dbz linker. Thus, its protection is required to avoid the accumulation of branched and acetylated side products and to better control chain extension.
The methylation of the Fmoc-Dbz amine in position 4 of MeDbz (FAA3166) renders this position less reactive and suppresses the formation of side products on the para-amino moiety.
Another Dawson linker derivative (FAA3168) is based on the allyloxycarbonyl (Alloc) protection of the Dbz amine in position 4. After successful synthesis, the Alloc group can quantitatively be removed under mild conditions, followed by thioester formation.
The derivative FAA3167 makes use of the tert-butyloxycarbonyl (Boc) group as an efficient and acid-labile protecting group for the deactivated Dbz amine. Unlike Alloc, the Boc group is removed by TFA during global deprotection of the peptide and cleavage from the resin without requiring an additional step.
Finally, the 4-amino-3-nitrobenzoic acid linker (FAA3169) was developed particularly for microwave-assisted peptide synthesis. After successful chain assembly, Dbz(NO2) can easily be converted into the Dbz linker by on-resin reduction with SnCl2. Even though epimerization of the C-terminal amino acid limits the use of the nitro derivative to the synthesis of peptide-Gly-thioesters, it suppresses the formation of side-products which are otherwise observed during microwave-assisted synthesis when using Dbz or Dbz(Alloc).
➔ Are you using the Fmoc-MeDbz-OH Dawson Linker in large scale on a regular basis? A robust and economic process allows us to promptly provide you with kg quantities at competitive prices and in high quality.
➔ Interested in more information on Native Chemical Ligation? ➔ Please feel free to download our new brochure on Ligation Technologies.
References:
A Reversible Protection Strategy To Improve Fmoc-SPPS of Peptide Thioesters by the N-Acylurea Approach; Santosh K. Mahto, Cecil J. Howard, John C. Shimko, and Jennifer J. Ottesen; ChemBioChem 2011; 12: 2488-2494. https://doi.org/10.1002/cbic.201100472.
An efficient Fmoc-SPPS approach for the generation of thioester peptide precursors for use in native chemical ligation; Blanco-Canosa, J. B. & Dawson, P. E.; Angew. Int. Ed. 2008; 47: 6851-6855. https://doi.org/10.1002/anie.200705471.
Protein Synthesis Assisted by Native Chemical Ligation at Leucine; Z. Harpaz, P. Siman, K. S. A. Kumar and A. Brik; Chembiochem : a European journal of chemical biology 2010; 11: 1232-1235. https://doi.org/10.1002/cbic.201000168.
A Semisynthesis Platform for Investigating Structure−Function Relationships in the N-Terminal Domain of the Anthrax Lethal Factor; B. L. Pentelute, A. P. Barker, B. E. Janowiak, S. B. H. Kent and R. J. Collier; ACS Chemical Biology 2010; 5: 359-364. https://doi.org/10.1021/cb100003r.
Alternative chemistries for the synthesis of thrombospondin-1 type 1 repeats; T. K. Tiefenbrunn, J. Blanco-Canosa and P. E. Dawson; Peptide Science 2010; 94: 405-413. https://doi.org/10.1002/bip.21486.
Solid-Phase Synthesis of Head to Side-Chain Tyr-Cyclodepsipeptides Through a Cyclative Cleavage From Fmoc-MeDbz/MeNbz-resins; G. A. Acosta, L. Murray, M. Royo, B. G. de la Torre, F. Albericio; Frontiers in Chemistry 2020; 8: 1-9. https://doi.org/10.3389/fchem.2020.00298.
Side-Chain Tyr-Cyclodepsipeptides Through a Cyclative Cleavage From Fmoc-MeDbz/MeNbz-resins; G. A. Acosta, L. Murray, M. Royo, B. G. de la Torre, F. Albericio; Chem. 2020; 8: 298. https://doi.org/10.3389/fchem.2020.00298.
Solid-phase synthesis of homodetic cyclic peptides from Fmoc-MeDbz-resin; S. A. H. A. Monaim, G. A. Acosta, M. Royo, A. El-Faham, B. G. de la Torre, F. Albericio; Tetrahedron Lett. 2018; 59: 1779-1782. https://doi.org/10.1016/j.tetlet.2018.03.084.