Welcome to Iris Biotech
For better service please confirm your country and language we detected.

For better service please confirm your country and language we detected.

Thank you very much for your interest in our products. All prices listed on our website are ex-works, Germany, and may attract customs duties when imported.
You may/will be contacted by the shipping company for additional documentation that may be required by the US Customs for clearance.
We offer you the convenience of buying through a local partner, Peptide Solutions LLC who can import the shipment as well as prepay the customs duties and brokerage on your behalf and provide the convenience of a domestic sale.
Continue to Iris Biotech GmbHSend request to US distributorPublished on 13/07/2021
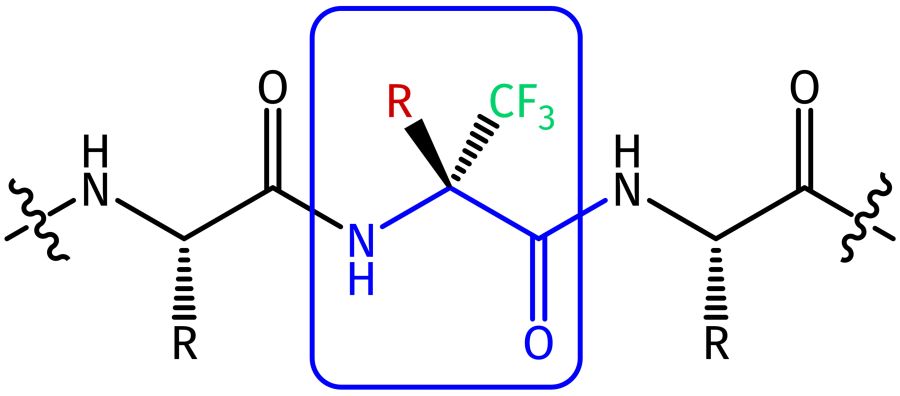
The demand for drug derivatives with improved pharmacokinetic and physicochemical profiles, including prolonged lifetime and proteolytic stability, as well as increased activity and lipophilicity drives research on innovative building blocks. In medicinal chemistry, approximately 20% of all approved pharmaceuticals have at least one fluorine atom present - with increasing tendency. Especially the introduction of trifluoromethyl groups into organic molecules can dramatically improve the above-mentioned properties. The CF3 group can be attached to aromatic rings of Phe, Trp, and Tyr, added to functional groups such as thiols, hydroxyls or amines, or be formed by replacing the hydrogen atoms in methyl groups.
The carbon-fluorine bond is characterized by a special hydrophobic character with a high dipole moment, described as polar hydrophobicity. The high electronegativity of fluorine impacts synthetic routes of any fluorine-containing molecule. Its electron withdrawing effect leads to high electron deficiency and thus decreased nucleophilicity at the neighboring amino nitrogen and, in the case of serine, also at the hydroxy oxygen. Thus, no more protection at these positions during any standard electrophilic coupling procedures during peptide synthesis is required. Yet, acetylation requires even harsher conditions.
Due to this altered chemical behavior and unique properties, the production of trifluoromethyl derivatives requires specific know-how and experience. An easy and convenient way, therefore, is the use of readily available fluorinated amino acids and protected dipeptide fragments, which easily can be incorporated in standard Fmoc/tBu synthesis protocols in automated or manual SPPS.
➔ Please inquire for the Trifluoromethyl-dipeptide and amino acid building block of your choice.
Besides, trifluoromethylated derivatives are characterized by their increased sterical requirement, impacting their conformational behavior. Both electronic and steric reasons render a cis-amide bond in α‑trifluoromethyl amino acids more favorable, inducing the formation of β-turns. In the case of the synthesis of cyclic peptides, cyclization yields can thereby be increased.
In addition, as fluorine is typically not found in natural peptides, 19F NMR spectroscopy serves as a sensitive and powerful tool facilitating the detection of even minor differences in the environment of the label.
Therefore, alpha-trifluoromethyl substituted amino acids are highly attractive building blocks for the design of biologically active compounds.
References:
Synthesis and Incorporation of alpha-Trifluoromethyl-Substituted Amino Acids into Peptides; B. Koksch, N. Sewald, H.-D. Jakubke, K. Burger; ACS Symposium Series 1996; 639. https://doi.org/10.1021/bk-1996-0639.ch003.
Approaches to Obtaining Fluorinated alpha-Amino Acids; J. Moschner, V. Stulberg, R. Fernandes, S. Huhmann, J. Leppkes, B. Koksch; Chem. Rev. 2019; 119(18): 10718-10801. https://doi.org/10.1021/acs.chemrev.9b00024.
What does fluorine do to a protein? Thermodynamic, and highly-resolved structural insights into fluorine-labelled variants of the cold shock protein. H. Welte, T. Zhou, X. Mihajlenko, O. Mayans, M. Kovermann; Sci Rep 2020; 10:2640. https://doi.org/10.1038/s41598-020-59446-w.
Expanding Organofluorine Chemical Space: The Design of Chiral Fluorinated Isosteres Enabled by I(I)/I(III) Catalysis; S. Meyer, J. Häfliger, R. Gilmour; Chem. Sci. 2021; https://doi.org/10.1039/D1SC02880D.
Fluorinated amino acids in protein design and engineering; N. C. Yoder, K. Kumar; Chem. Soc. Rev. 2002; 31(6): 335-341. https://doi.org/10.1039/b201097f.
Fluorinated amino acids: compatibility with native protein structure and effects on protein-protein interactions; M. Salwiczek, E. K. Nyakatura, U. I. M. Gerling, S. Ye, B. Koksch; Chem. Soc. Rev. 2012; 41: 2135-2171. https://doi.org/10.1039/C1CS15241F.
Peptide modification by incorporation of alpha-trifluoromethyl substituted amino acids; B. Koksch, N. Sewald, K. Burger, H.-D. Jakubke; Amino Acids 1996; 11: 425-434. https://doi.org/10.1007/BF00807946.